Sham surgery trials have unveiled startling truths about some of the most widely adopted surgical procedures, suggesting that in certain cases, their efficacy is on par with that of a placebo. This revelation has cast a critical spotlight on the rigorous evaluation of medical devices and surgical techniques, particularly those aimed at addressing complex health issues like obesity and joint pain. The history of intragastric balloons, once heralded as a breakthrough in weight management, serves as a potent case study in the challenges of bringing novel medical technologies to market and the critical importance of independent, robust clinical investigation.
The Rise and Fall of the Gastric Balloon: A Cautionary Tale
Intragastric balloons, first introduced with significant fanfare in the 1980s, were designed to offer a less invasive approach to weight loss. The concept was straightforward: a balloon, filled with air or water, would be implanted into the stomach, mechanically occupying space and thereby inducing a sensation of fullness, leading to reduced food intake. This innovation promised a solution for individuals struggling with obesity who sought an alternative to more drastic bariatric surgeries.
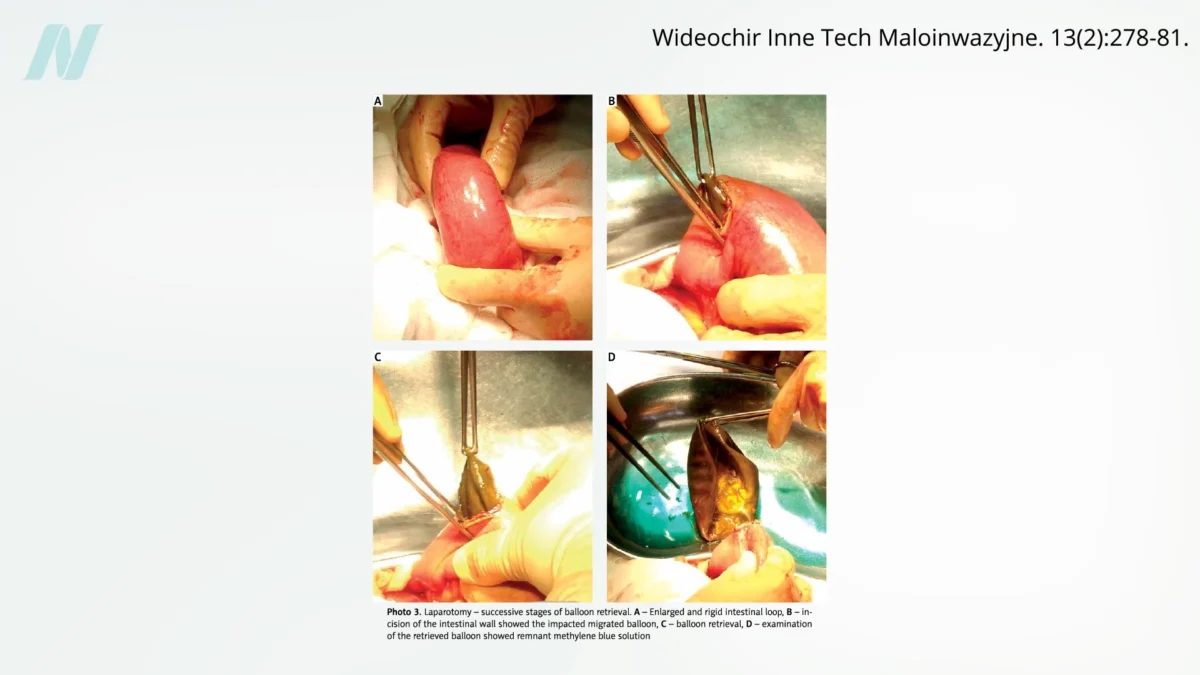
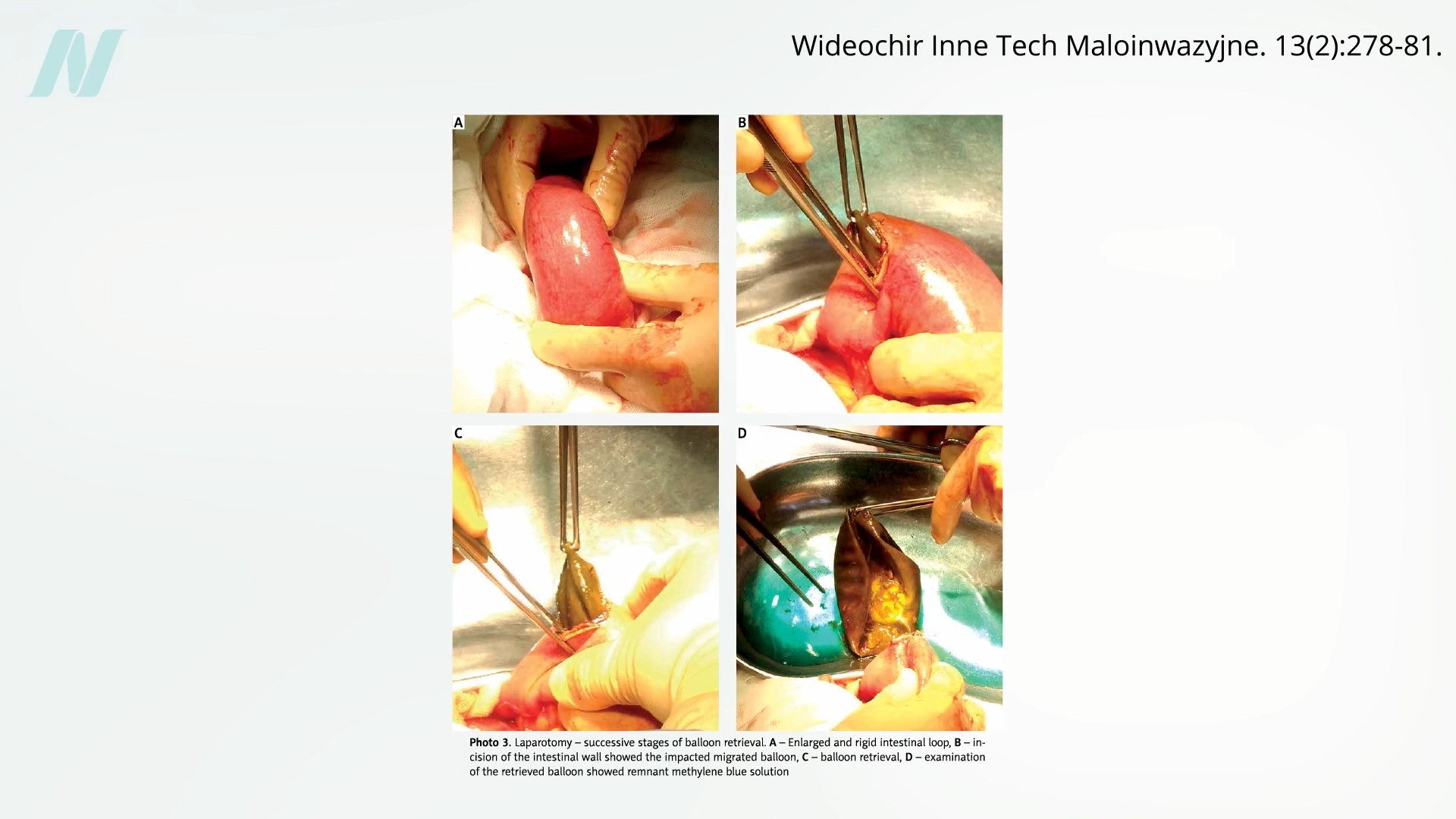
However, the trajectory of intragastric balloons underscores a recurring issue in the medical device industry: the premature introduction of products into the market before comprehensive evidence of their effectiveness and safety has been established. Early studies and clinical observations began to reveal significant shortcomings. A pivotal study conducted at the Mayo Clinic, for instance, exposed a critical flaw in the original balloon designs. The findings indicated that a staggering 8 out of 10 balloons spontaneously deflated. This malfunction presented a serious and potentially life-threatening complication: the deflated balloon could migrate into the intestines, leading to a severe bowel obstruction. Such an event necessitates urgent medical intervention and can have devastating consequences.
Beyond the risk of mechanical failure, the intragastric balloons of that era were also associated with significant gastrointestinal distress. Reports indicated that approximately half of the patients who received the balloons experienced gastric erosions, a condition involving damage to the stomach lining. This damage could manifest as pain, bleeding, and other serious complications.
Perhaps the most damning evidence against the early iterations of intragastric balloons emerged when their efficacy in promoting sustainable weight loss was rigorously assessed. When compared to established, non-surgical methods such as structured diets and behavioral modification programs, the balloons failed to demonstrate a significant or lasting advantage. The incremental weight loss achieved, if any, often did not justify the risks and discomfort associated with the device. Consequently, the initial wave of intragastric balloons was eventually withdrawn from the market, a testament to their insufficient performance and concerning safety profile.

The Resurgence of Intragastric Balloons: New Devices, Lingering Concerns
Following a hiatus of over three decades, intragastric balloons re-entered the U.S. market after the Food and Drug Administration (FDA) began approving new generations of these devices in 2015. This reintroduction coincided with the implementation of the Sunshine Act, a landmark piece of legislation enacted to increase transparency in financial relationships between healthcare providers and the pharmaceutical and medical device industries. The Act mandates the disclosure of payments made by these entities to physicians, aiming to shed light on potential conflicts of interest.
While the Sunshine Act has been instrumental in revealing the extent of financial ties between doctors and industry, a less appreciated aspect pertains to the payments surgeons can receive from device manufacturers. Data from various reports indicate that top physician recipients of industry payments can receive millions of dollars annually from device companies. This financial leverage raises critical questions about the impartiality of surgical recommendations and the reporting of clinical trial outcomes. A concerning trend observed has been the minority of published studies that adequately disclose these potential conflicts of interest, potentially skewing the perception of a device’s true benefits.
The newer intragastric balloons, while designed with improved safety features and materials, are not without their own set of risks. Although they are considered reversible, a significant advantage over permanent bariatric surgeries, their implantation is far from benign. The FDA has issued multiple advisories detailing the potential complications, which, in the most severe cases, can include death.
One of the most perplexing and dangerous complications associated with intragastric balloons is the risk of stomach perforation. While the balloons are typically smooth and rounded, they can induce severe and persistent nausea and vomiting. This forceful and sustained expulsion of stomach contents can, in extreme instances, lead to a rupture of the stomach wall, a life-threatening emergency. Nausea and vomiting are widely acknowledged as very common side effects, affecting a substantial majority of patients who undergo balloon placement. Furthermore, the persistent vomiting can contribute to severe nutrient deficiencies, potentially leading to long-term health problems.
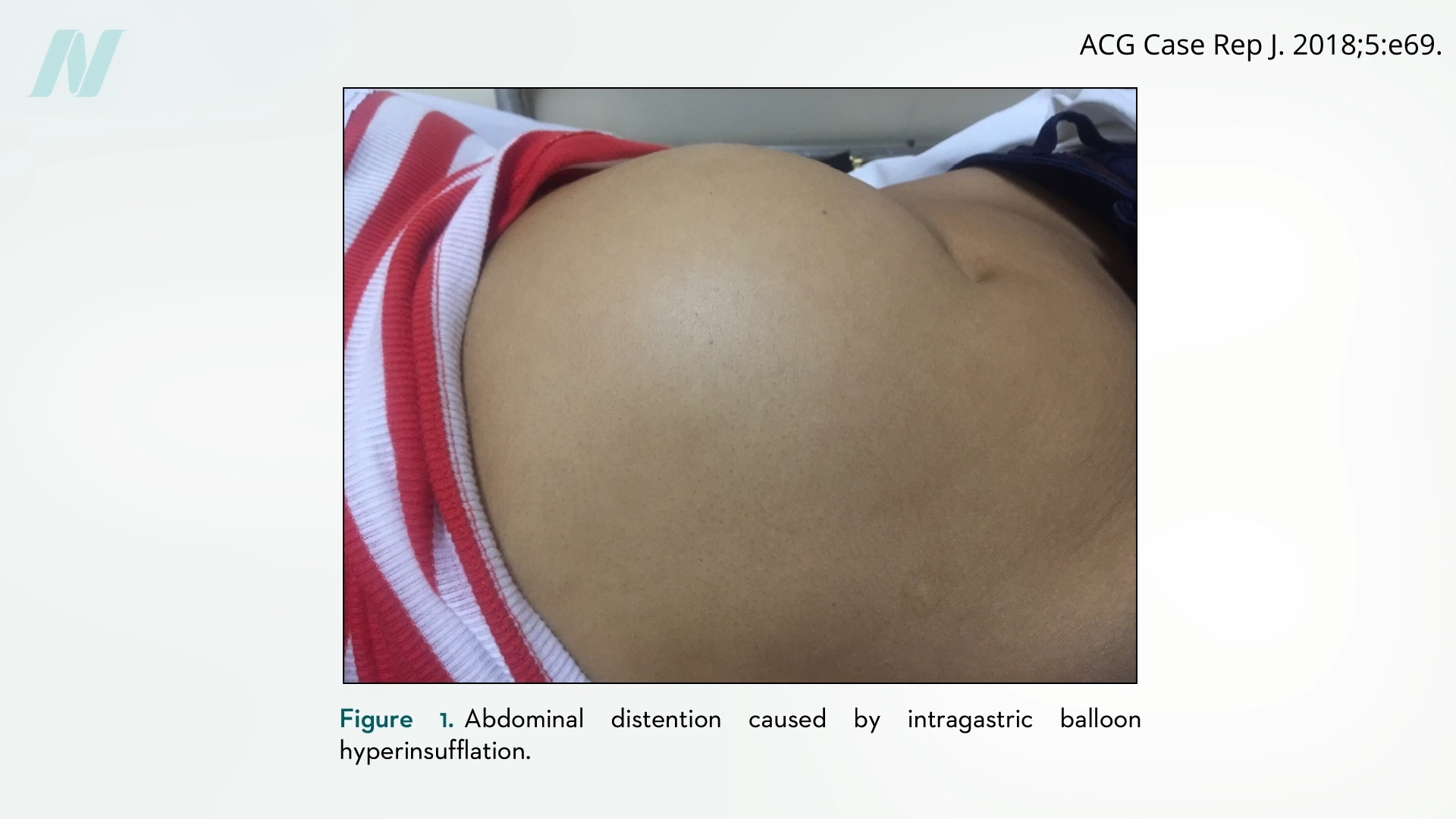
Complications arising from intragastric balloons can be broadly categorized into two main issues: those related to deflation and those linked to overinflation. As seen with the earlier models, deflation can lead to the balloon fragmenting or migrating, causing potentially fatal bowel obstructions. Conversely, some newer devices have been associated with unexpected overinflation. This phenomenon can cause significant pain, intense vomiting, and severe abdominal distention, requiring immediate medical attention.
Intriguingly, the issue of spontaneous overinflation in implanted medical devices is not entirely novel. It has been documented in the context of breast implants, where reports of "spontaneously autoinflating breast implants" have emerged. These implants have been observed to increase breast volume by over 50% without external intervention, a phenomenon that remains poorly understood and underreported. Historically, some of the earliest experimental intragastric balloons were, in fact, adapted from designs initially used for breast implants, hinting at a shared underlying mechanical vulnerability.
The Unsettling Power of the Placebo Effect: Insights from Sham Surgery Trials
The debate surrounding the effectiveness of intragastric balloons, and indeed many other surgical procedures, is significantly informed by the concept of the placebo effect and the rigorous methodology of sham surgery trials. In the realm of drug development, randomized controlled trials (RCTs) often compare a new medication against a placebo (an inert substance) to isolate the drug’s true therapeutic effect. However, applying this principle to surgery presents unique ethical and logistical challenges.
To address this, researchers have employed "sham surgery" as a control group in certain surgical trials. In a sham surgery, patients undergo a procedure that mimics the operative steps of the actual surgery, including incisions and the manipulation of tissues, but without performing the definitive therapeutic intervention. For example, in a sham knee surgery, a patient might have their knee surgically opened and saline solution splashed onto the joint, simulating the appearance of arthroscopic surgery, but without any actual tissue debridement or repair.
One of the most impactful sham surgery trials was published in The New England Journal of Medicine in 2002, focusing on arthroscopic knee surgery, a procedure performed millions of times annually worldwide for conditions like osteoarthritis and knee injuries. Billions of dollars are spent on this surgery, which involves inserting a small camera and instruments into the knee joint to trim damaged cartilage. The study randomized patients with knee pain into three groups: one receiving arthroscopic debridement, another undergoing arthroscopic lavage (washing out the joint), and a third group receiving a sham surgery.
The results of this trial sent shockwaves through the orthopedic community. While patients in both the debridement and lavage groups reported improvements in their knee pain and function, the patients who underwent the sham surgery experienced nearly identical outcomes. This finding strongly suggested that the perceived benefits of arthroscopic knee surgery were largely attributable to the placebo effect, the power of suggestion, and the natural healing processes, rather than the surgical intervention itself. The implications were profound, questioning the value of a procedure that was a cornerstone of orthopedic practice and a significant revenue generator. The trial ignited a fierce debate about the ethics of performing sham surgeries, with some professional medical associations questioning the morality of surgeons and the judgment of patients who consented to such trials.
This groundbreaking study has paved the way for similar investigations into other common surgical procedures. Rotator cuff shoulder surgery, another widely performed intervention for shoulder pain and dysfunction, is currently facing a similar crisis of confidence, with emerging evidence suggesting that sham-controlled trials may reveal a significant placebo component to its effectiveness.
Evaluating the Evidence: Industry Funding and the Challenge of Objectivity
The efficacy of intragastric balloons has also been scrutinized through the lens of sham-controlled trials. When subjected to this rigorous methodology, both older and newer generations of balloons have frequently failed to demonstrate a statistically significant weight-loss benefit over sham procedures. Even in instances where some weight loss is observed, it is often temporary. This is partly due to the fact that balloons are typically intended for short-term use, often limited to six months, due to escalating risks of deflation and associated complications. Attempts to extend the duration of balloon use or to replace deflated balloons with new ones have generally not yielded improved long-term weight management outcomes in sham-controlled studies.
The transient nature of any observed effects on appetite and satiety is another critical finding. As the body adapts to the presence of the balloon, the initial sensations of fullness and reduced hunger may diminish, leading to a return to previous eating habits. This suggests that the physiological adaptations induced by the balloon may be short-lived.
The broader implications of sham surgery research extend beyond specific interventions. They highlight a systemic challenge within modern medicine: the potential for financial incentives to influence clinical practice and research outcomes. While the medical profession rightly champions evidence-based practice and often criticizes the spread of misinformation and the rejection of scientific consensus, as seen in anti-vaccination movements, these sham surgery studies reveal a parallel vulnerability within medicine itself. The notion that some widely performed and profitable surgeries may be ineffective, or even detrimental, echoes the concept of "fake news" and "alternative facts" within the scientific community.
The Path Forward: Transparency, Rigor, and Patient-Centered Care
The findings from sham surgery trials and critical analyses of medical devices like intragastric balloons underscore the urgent need for enhanced transparency, rigorous independent research, and a renewed focus on patient-centered decision-making. Patients considering any surgical intervention, particularly those for elective purposes or chronic conditions, should be empowered with comprehensive, unbiased information regarding the risks, benefits, and evidence supporting the procedure.
The medical industry, including pharmaceutical companies and medical device manufacturers, must be held to the highest standards of ethical conduct and scientific integrity. Disclosure of financial relationships between industry and healthcare providers is a crucial step, but it must be coupled with a commitment to conducting and publishing high-quality, independent research that prioritizes patient well-being over commercial interests.
Moving forward, the medical community must embrace a culture of continuous re-evaluation and a willingness to challenge established practices when evidence dictates. This includes investing in long-term, unbiased studies that not only assess immediate outcomes but also track the durability of results and potential late-onset complications. The ultimate goal is to ensure that surgical interventions, and indeed all medical treatments, are not only safe and effective but also offer genuine value and improved quality of life for patients.
The next installment in this series will delve into other extreme weight-loss devices and their purported efficacy. For those seeking further information on bariatric surgery and related topics, a comprehensive review of related posts is recommended. Furthermore, resources such as the book How Not to Diet, which focuses on sustainable weight loss strategies, are available through libraries and booksellers, with all proceeds dedicated to charitable causes.