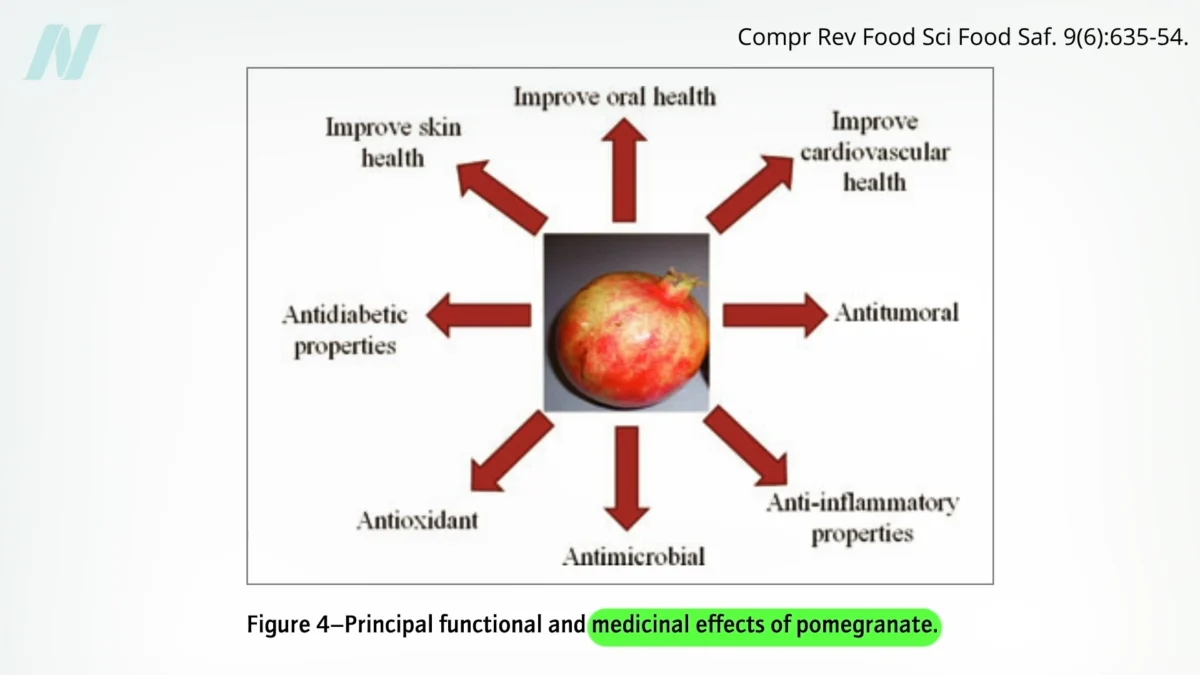
The potential health benefits of pomegranates have long been a subject of both scientific inquiry and commercial promotion. While the fruit and its juice are lauded for their antioxidant properties and purported ability to combat a range of ailments, including weight gain, diabetes, chronic obstructive pulmonary disease (COPD), prostate cancer, osteoarthritis, and rheumatoid arthritis, a closer examination of the scientific literature and regulatory actions reveals a more nuanced picture. The journey of pomegranate product claims from the laboratory to the consumer has been marked by legal battles, scientific rigor, and the ongoing challenge of translating in-vitro findings into tangible human health outcomes.
The POM Wonderful Legal Saga: A Landmark Case for Health Claims
A significant turning point in the public perception and marketing of pomegranate products was the protracted legal battle between POM Wonderful and the Federal Trade Commission (FTC). This case, which ultimately reached the U.S. Court of Appeals and was denied review by the Supreme Court, centered on advertising claims made by POM Wonderful regarding the health benefits of its pomegranate juice. The FTC argued that many of these advertisements presented misleading or unsubstantiated claims about the product’s ability to treat, prevent, or reduce the risk of serious diseases.
In its ruling, a panel of judges concluded that numerous POM Wonderful advertisements had indeed made false or misleading claims. The company had aggressively promoted its products, citing various medical studies to suggest that daily consumption could address conditions such as heart disease, prostate cancer, and erectile dysfunction. However, the court emphasized that such claims, particularly those related to treating or preventing disease, require robust scientific evidence. Citing the principles of the U.S. First Amendment, which does not extend protection to deceptive advertising, the court delivered a significant blow to the company and its owners.
The landmark decision mandated that to substantiate claims of treating or preventing disease, at least one randomized clinical trial would be required. This ruling set a new standard for health-related marketing, emphasizing the necessity of human trials that meet rigorous scientific criteria before broad health benefits can be advertised. The FTC’s victory underscored the importance of consumer protection and the need for scientific evidence to back up health-related marketing assertions.

Decoding the Pomegranate: From Lab Bench to Human Trials
While the legal battles highlighted the challenges in substantiating broad health claims, the scientific community continues to investigate the complex phytochemicals present in pomegranates. Reviews of the medical literature often showcase diagrams illustrating a wide array of potential medicinal effects, frequently highlighting the fruit’s potent antioxidant and anti-inflammatory compounds. These initial findings, often derived from laboratory studies (in vitro), have fueled much of the public interest and commercial promotion.
However, a deeper dive into the research reveals that many of these promising initial findings do not translate directly to human health benefits. For instance, some early research pointing to "antiobesity effects" was based on studies conducted on mouse models using pomegranate leaf extract. Such findings, while scientifically interesting, are not directly applicable to human consumption patterns, as consumers typically ingest the fruit’s arils or juice, not its leaves, and the physiological responses of mice differ significantly from those of humans.
Examining the Evidence: Weight Management, Cardiovascular Health, and Beyond
The efficacy of pomegranate consumption for weight management in humans has been a particular area of scrutiny. A comprehensive review of randomized controlled clinical trials—the gold standard for human research—has indicated that pomegranates do not have a significant impact on body weight, body mass index (BMI), abdominal fat, or overall body fat percentage. This suggests that while pomegranates may offer other benefits, they are unlikely to be a standalone solution for weight loss.
Similarly, claims regarding the prevention and treatment of cardiovascular diseases have faced rigorous scientific examination. An extensive review of 25 clinical trials, encompassing metrics such as cholesterol levels, blood pressure, arterial function, atherosclerotic plaque formation, and platelet function, failed to yield significant indications of benefit. Even in the most well-designed studies, the evidence supporting a direct cardioprotective role for pomegranate consumption remained inconclusive.
POM Wonderful itself funded a study investigating the effects of pomegranate juice on erectile dysfunction, but this research also failed to demonstrate a significant benefit. Further studies have explored the impact of pomegranates on diabetes markers, with findings indicating no discernible benefit. Likewise, research into chronic obstructive pulmonary disease (COPD) has not found evidence of improvement with pomegranate consumption.
Antioxidant Power: In Vitro vs. In Vivo Realities
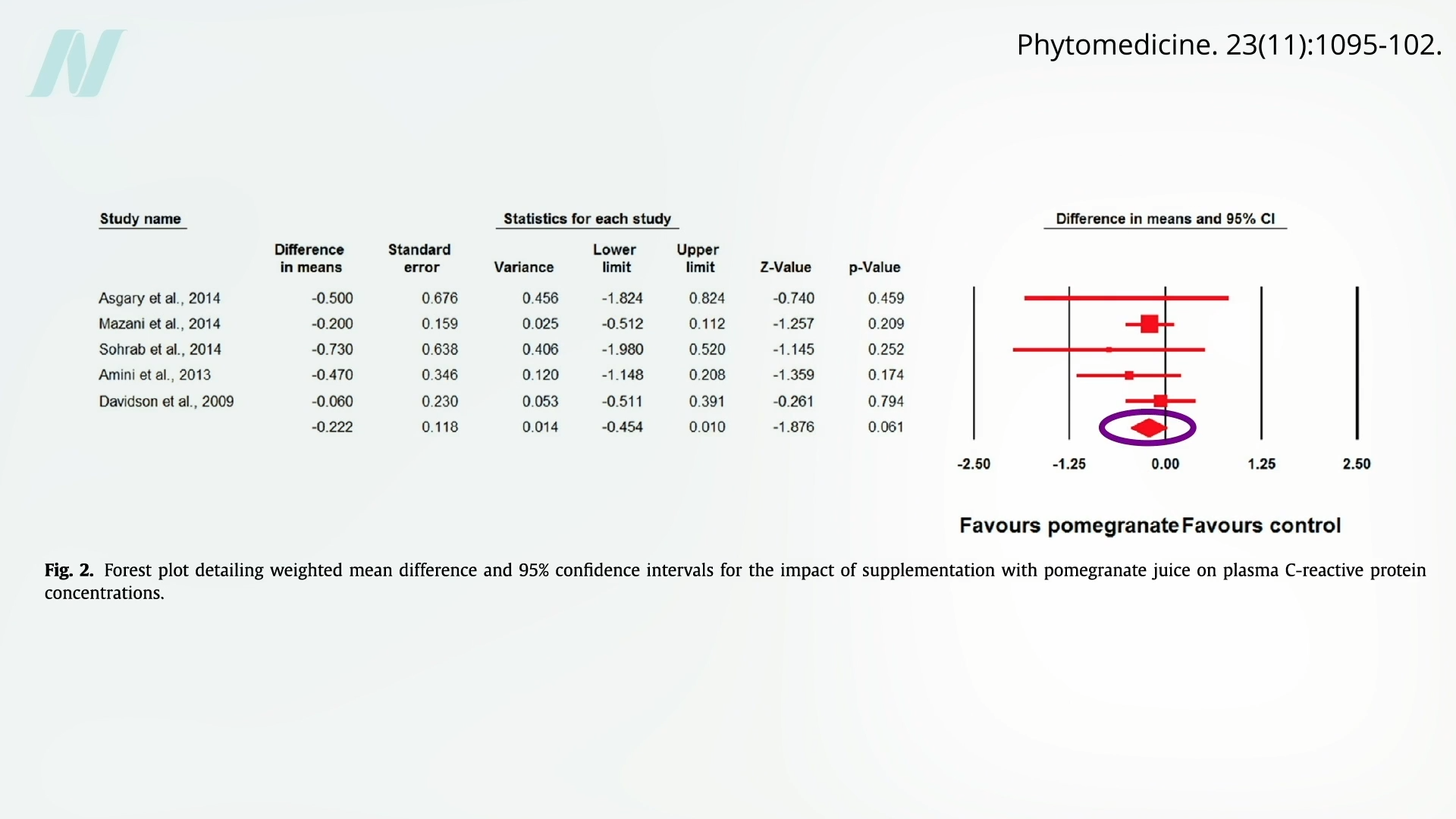
Much of the initial excitement surrounding pomegranates stemmed from their high antioxidant capacity, observed in laboratory settings (in vitro). Antioxidants are known to combat oxidative stress, a process linked to aging and various diseases. However, the effectiveness of these antioxidants once ingested by humans (in vivo) is a critical question.
A meta-analysis of data from 11 randomized controlled trials revealed that pomegranate intake did not significantly increase the total antioxidant capacity in the bloodstream. This finding suggests that some of the most potent antioxidants present in pomegranates may not be effectively absorbed into the human body. Consequently, this lack of bioavailability could explain why pomegranate supplementation did not appear to influence oxidative stress markers in tissue samples from prostate cancer patients. The critical question, of course, remains whether it impacts the cancer itself.
Prostate Cancer Research: Promising Beginnings, Unfulfilled Expectations
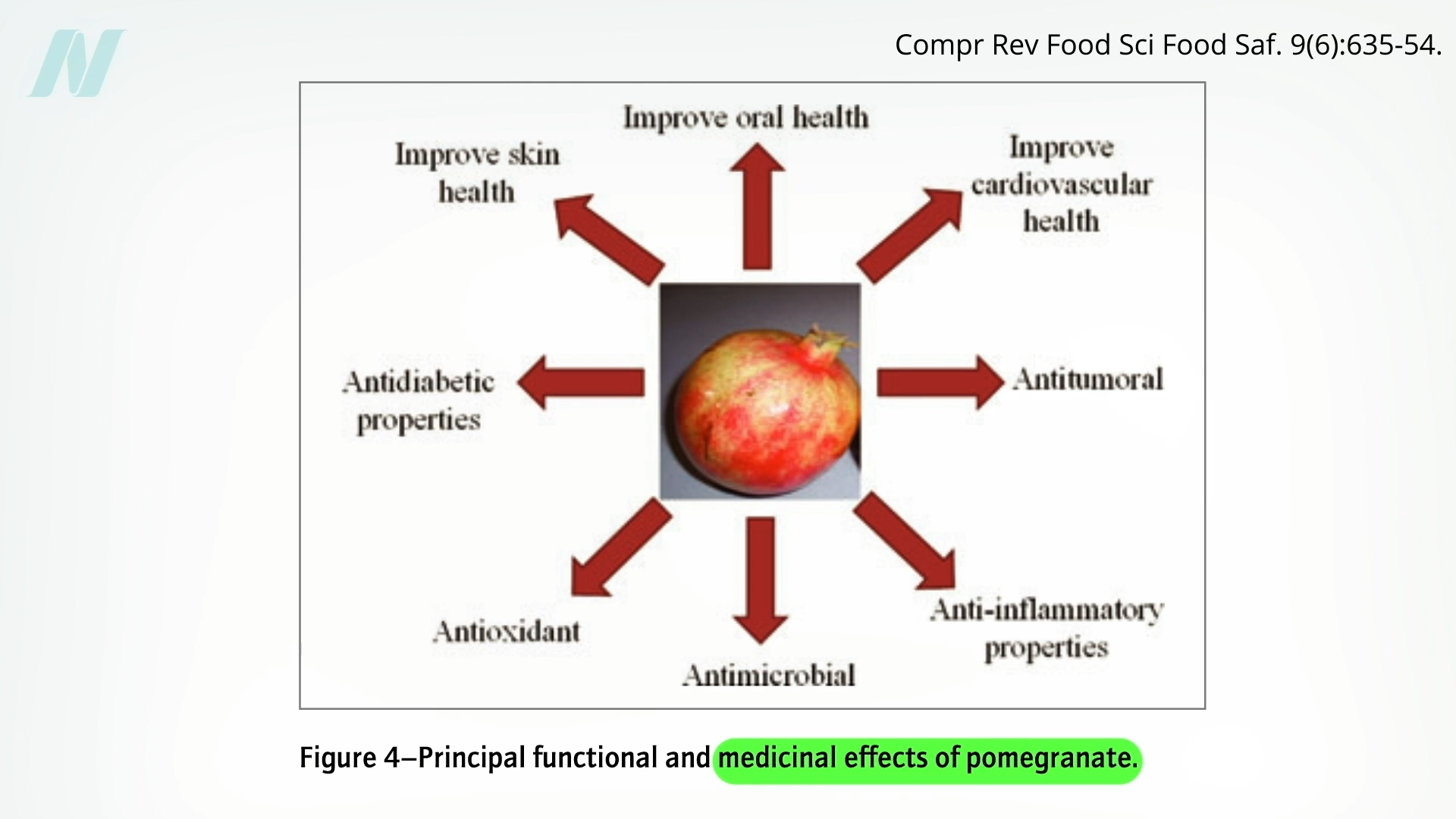
The potential anticancer activity of pomegranates has generated considerable interest, particularly concerning prostate cancer. Early research had shown promise, but subsequent, more rigorous testing has failed to consistently confirm these initial findings. For example, a randomized controlled trial examining daily pomegranate intake found no impact on prostate-specific antigen (PSA) levels, a key indicator of prostate cancer progression.
Another randomized, double-blind, placebo-controlled trial also revealed essentially no difference in prostate cancer disease progression between those who consumed pomegranate and those who received a placebo. These findings underscore the gap between preliminary observations and definitive clinical outcomes.
Inflammation and Joint Health: A Shifting Landscape of Evidence
The anti-inflammatory properties of pomegranate have also been a focus of research, with potential implications for inflammatory diseases. Early studies demonstrated that pomegranate extract could exhibit cartilage-protective effects in laboratory settings when applied to human osteoarthritic cartilage samples. These in-vitro results showed that inflammation significantly increased cartilage damage, but this damage appeared to be mitigated by increasing amounts of pomegranate extract.
However, translating these petri-dish findings into human benefits requires demonstrating that the active components of pomegranate are bioavailable and reach the affected joints. While some studies in rabbits suggested bioavailability of cartilage-protecting compounds, raising hopes for a safe and non-toxic treatment option for osteoarthritis, the clinical evidence has been less conclusive.
The first clinical trial on pomegranate juice and osteoarthritis was not placebo-controlled, and while participants drinking the juice reported less stiffness and impairment, the improvements were not statistically significant compared to doing nothing. This highlights the critical need for robust control groups in research design.
Further investigation into rheumatoid arthritis also encountered initial challenges. A study titled "Consumption of Hydrolyzable Tannins-Rich Pomegranate Extract Suppresses Inflammation and Joint Damage in Rheumatoid Arthritis" was conducted by the POM company and used a mouse model, again raising questions about direct human applicability. An open-label study involving eight rheumatoid arthritis patients showed reduced tender joints in those who consumed pomegranate extract, but the lack of a control group limited the conclusions that could be drawn.
The Turning Point: Rigorous Trials Yield More Definitive Results
The landscape of evidence began to shift with the advent of well-designed, randomized, double-blind, placebo-controlled trials. One such study involved half of the participants unknowingly receiving pomegranate extract and the other half receiving a placebo. This research indicated significant improvements in various markers of rheumatoid arthritis, including the number of swollen and tender joints, pain intensity, disease activity scores, and morning stiffness, when compared to the placebo group. These findings suggest that pomegranate extracts may indeed offer therapeutic benefits for individuals with rheumatoid arthritis.
However, the mode of delivery remains a consideration. While pomegranate extracts showed promise, the question of whether to consume the whole fruit or supplements arises. Laboratory testing of 19 pomegranate supplements revealed a concerning issue: the majority did not appear to contain any actual pomegranate. Only a small fraction of the tested supplements were found to be authentic, highlighting potential quality control and product integrity challenges in the supplement market. This underscores the importance of relying on whole foods when possible, or exercising extreme caution and due diligence when choosing supplements.
Broader Implications and Future Directions
The scientific journey of pomegranates illustrates a critical principle in health research: the distinction between preliminary laboratory findings and clinically proven human benefits. While the potent antioxidant and anti-inflammatory compounds in pomegranates hold theoretical promise, their actual impact on human health requires rigorous, well-controlled studies.
The legal precedent set by the POM Wonderful case serves as a vital reminder for both manufacturers and consumers. For manufacturers, it emphasizes the ethical and legal obligation to substantiate health claims with robust scientific evidence, particularly through human clinical trials. For consumers, it encourages a critical approach to health-related marketing, urging them to look beyond anecdotal evidence and promotional materials to seek out scientifically validated information.
The ongoing research into pomegranates, particularly in areas like rheumatoid arthritis, suggests that the fruit and its derivatives may indeed offer tangible health benefits. However, continued investigation is crucial to fully understand the mechanisms of action, optimal dosages, and long-term effects. As scientific understanding evolves, the narrative surrounding pomegranates is likely to become more refined, distinguishing between scientifically supported benefits and unsubstantiated claims, ultimately empowering consumers to make informed choices about their health.