The intricate lining of our intestines, a formidable barrier essential for health, is far more vulnerable to disruption than commonly understood. Emerging scientific evidence points to a wide array of everyday substances, from common medications to dietary staples, as significant contributors to the breakdown of this critical defense mechanism, a condition often referred to as "leaky gut" or increased intestinal permeability. This newfound understanding positions intestinal permeability not merely as a symptom, but as a potential nexus for both the prevention and treatment of a spectrum of diseases.
The sheer scale of the intestinal barrier underscores its importance. Composed of a single layer of epithelial cells, folded into villi and microvilli, it boasts a surface area exceeding 4,000 square feet – a vast expanse larger than a professional tennis court. Maintaining this immense, highly specialized tissue demands a substantial portion of the body’s resources, estimated to consume approximately 40% of its total energy expenditure. This significant investment highlights the vital role the intestinal barrier plays in nutrient absorption, immune regulation, and preventing the ingress of harmful substances into the bloodstream.
The Science of Intestinal Permeability
Intestinal permeability refers to the degree to which the intestinal lining allows substances to pass through it. In a healthy gut, this barrier is tightly regulated, permitting the passage of digested nutrients while meticulously blocking toxins, pathogens, and undigested food particles. However, when this barrier integrity is compromised, it becomes "leaky," allowing undesirable molecules to traverse into the systemic circulation, triggering inflammatory responses and potentially contributing to a wide range of health issues.
Researchers have developed methods to quantify intestinal permeability. One notable technique involves the administration of inert, non-absorbable dyes, such as blue food coloring. In individuals with a healthy intestinal barrier, these dyes are expected to remain within the digestive tract and be excreted. Conversely, in individuals with significantly compromised gut integrity, such as those suffering from severe sepsis, these dyes have been detected in the bloodstream. This stark difference underscores the direct correlation between gut barrier health and systemic well-being.
Common Culprits Behind a Compromised Gut
The notion that a "leaky gut" is solely the domain of critically ill patients in intensive care units is a misconception. The findings suggest that even seemingly benign, over-the-counter medications can inflict damage. Non-steroidal anti-inflammatory drugs (NSAIDs), a class of widely used pain relievers, have been identified as significant disruptors of intestinal barrier function.
NSAID Medications and Their Impact
Studies have demonstrated that taking even a single dose of common NSAIDs can increase intestinal permeability. For instance, research has shown that ingesting two standard 325 mg aspirin tablets, or two extra-strength 500 mg tablets, can acutely enhance gut leakiness. This effect is not limited to severe pain relief; even healthy individuals may experience gastrointestinal barrier dysfunction following NSAID use.
The concern extends to buffered aspirin, a formulation designed to mitigate gastrointestinal irritation. However, evidence suggests that buffering agents may offer limited protection. Studies using endoscopic examinations have revealed that both regular aspirin and buffered aspirin formulations can induce multiple erosions and redness within the stomach and intestinal lining. Remarkably, this damage can manifest rapidly, with observable changes occurring within as little as five minutes of administration. This rapid onset of damage highlights the immediate vulnerability of the gut lining to these medications.
Acetaminophen, commonly known as Tylenol in the United States, is often considered a safer alternative for pain relief with respect to gastrointestinal damage, provided individuals do not have pre-existing liver conditions. However, the picture is not entirely clear for all supplements. Paradoxically, some studies suggest that vitamin C supplements, rather than offering protection, may exacerbate aspirin-induced increases in gut leakiness. This interaction warrants further investigation to fully understand the implications for individuals taking both substances.
The implications of NSAID-induced gut leakiness extend beyond direct gastrointestinal discomfort. There is a growing body of evidence linking NSAIDs to an increased risk of food-induced anaphylaxis. These drugs are implicated in up to 25% of such severe allergic reactions. The proposed mechanism suggests that by increasing intestinal permeability, NSAIDs allow microscopic food particles to enter the bloodstream, where they can trigger an exaggerated immune response, leading to life-threatening anaphylactic episodes. This association suggests a more profound systemic impact of compromised gut barrier function, extending to immune hypersensitivity.
Strenuous Exercise and Gut Health
While often lauded for its health benefits, strenuous exercise can, under certain circumstances, temporarily impact intestinal integrity. An intense workout, such as an hour at 70% of maximum capacity, can divert significant blood flow away from internal organs, including the intestines, towards working muscles. This transient reduction in blood supply can lead to mild gut leakiness. This effect can be amplified if athletes concurrently use NSAIDs, a practice unfortunately common among individuals seeking to manage exercise-induced discomfort. The combination of physiological stress from intense exercise and the pharmacological insult of NSAIDs creates a synergistic effect that can further compromise the gut barrier.

Alcohol Consumption and Its Consequences
Alcohol is another dietary component identified as a risk factor for food allergy attacks, again through the mechanism of increased gut leakiness. Similar to NSAIDs, alcohol can compromise the integrity of the intestinal lining, allowing food particles to enter the circulation and potentially trigger allergic reactions. However, the gut’s capacity for repair is also noteworthy. Studies suggest that abstaining from alcohol can lead to the healing of the gut barrier, underscoring the reversible nature of some of these disruptions.
Dietary Fat and Gut Microbiota Imbalance
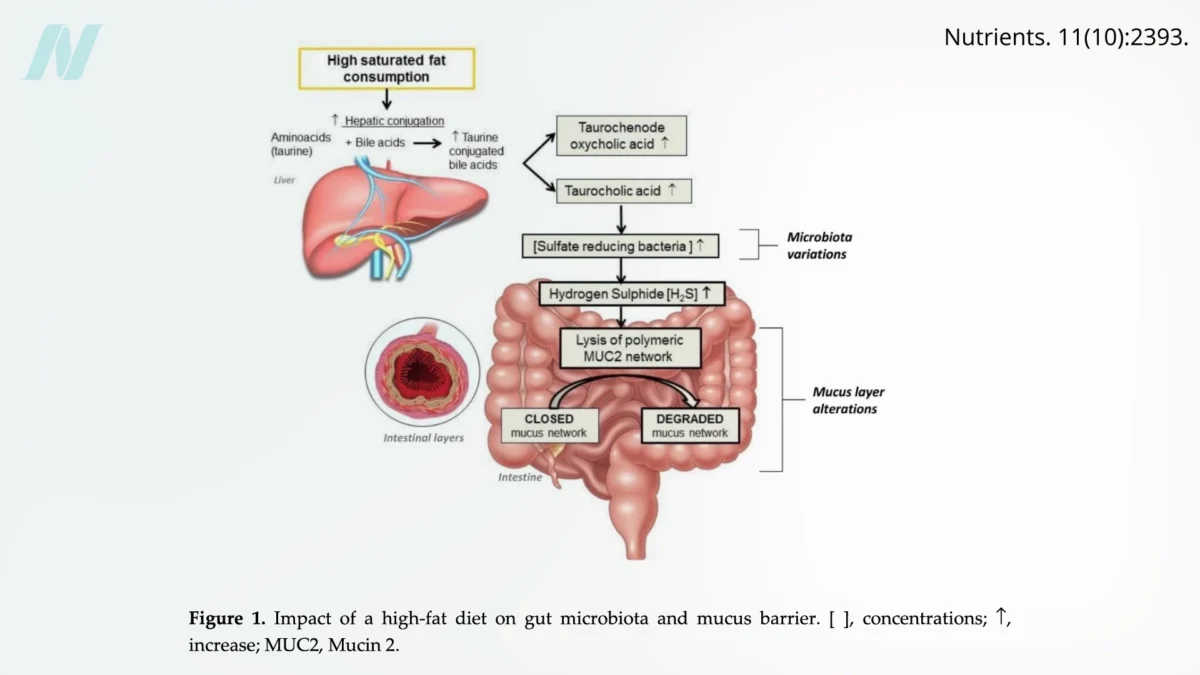
The impact of dietary choices on gut health is a rapidly evolving area of research. Elevated consumption of saturated fats, prevalent in red meat, dairy products, and many processed "junk" foods, has been linked to detrimental changes in the gut environment. Saturated fats can promote the growth of specific bacteria that produce hydrogen sulfide, a gas with a rotten-egg odor. This gas can degrade the protective mucus layer that lines the intestinal wall, diminishing its barrier function.
The broader implications of high-fat diets on intestinal health are significant. While much of the initial research was conducted on animal models or in vitro, human studies are beginning to solidify these findings. The increasing global prevalence of obesity and cardiometabolic disorders has coincided with a widespread shift from traditional, lower-fat diets to higher-fat Westernized diets. This dietary transition is strongly associated with alterations in the gut microbiome, the complex ecosystem of microorganisms residing in the intestines.
Research has indicated that a disturbed gut flora is linked to an increased risk of many chronic diseases, including those related to cardiometabolism. Rodent studies have consistently shown that high-fat diets can unbalance the microbiome and impair the gut barrier, leading to disease development. To bridge the gap between animal studies and human health, researchers have conducted interventional trials. A notable six-month randomized controlled feeding trial investigated the effects of dietary fat on the gut microbiota in humans. The findings were compelling: higher fat consumption was indeed associated with unfavorable shifts in the gut microbiome and increased levels of pro-inflammatory markers in the blood.
Interestingly, this trial demonstrated that the detrimental effects were not solely attributable to saturated fats. Simply replacing refined carbohydrates with refined fats – for example, substituting white rice and wheat flour with soybean oil – yielded significant negative outcomes. These results carry substantial implications for public health recommendations. Countries undergoing dietary transitions towards Westernized patterns are advised to caution against increasing dietary fat intake. Conversely, nations that have already adopted such diets may benefit from strategies aimed at reducing overall fat consumption.
A New Frontier in Disease Prevention and Therapy
The growing body of evidence linking intestinal permeability to various health conditions, including celiac disease and inflammatory bowel diseases (IBD), positions this physiological state as a critical area for further research and intervention. The ability to identify and modulate intestinal leakiness could unlock novel strategies for preventing the onset of chronic diseases and managing existing ones.
The precise mechanisms by which gut barrier dysfunction contributes to diseases like celiac and IBD are complex and multifactorial. In celiac disease, for example, a genetic predisposition combined with gluten ingestion leads to an immune response that damages the small intestine. However, research suggests that increased intestinal permeability may precede or exacerbate this damage, allowing gluten peptides to penetrate the barrier more readily and trigger a more robust immune reaction.
Similarly, in inflammatory bowel diseases such as Crohn’s disease and ulcerative colitis, a breakdown in the intestinal barrier is a hallmark. This leakiness allows gut microbes and their products to cross the intestinal wall, stimulating chronic inflammation and immune dysregulation. Understanding and potentially reversing this barrier defect could offer new therapeutic avenues for these debilitating conditions.
Broader Implications and Future Directions
The implications of these findings are far-reaching, suggesting a need for a re-evaluation of our dietary habits and medication use. While definitive treatment protocols are still emerging, the data points towards lifestyle modifications as a primary approach to supporting gut barrier health.
- Dietary Adjustments: Prioritizing whole, unprocessed foods, limiting saturated and refined fats, and moderating alcohol intake are likely to be key strategies. The focus on reducing refined carbohydrates in favor of healthier fat sources in the human trial highlights the nuanced relationship between macronutrient composition and gut health.
- Medication Prudence: A more cautious approach to NSAID use, particularly for individuals with a history of gastrointestinal issues or those prone to allergic reactions, may be warranted. Exploring alternative pain management strategies and consulting with healthcare professionals about the risks and benefits of NSAIDs is crucial.
- Exercise Moderation: While strenuous exercise offers numerous health advantages, individuals engaging in intense physical activity, especially when using NSAIDs, should be aware of the potential for temporary gut leakiness and discuss strategies with their healthcare providers.
Further research is imperative to fully elucidate the complex interplay between diet, medications, lifestyle, and intestinal barrier function. Large-scale, long-term human interventional trials are needed to confirm these findings and develop evidence-based guidelines for preventing and managing conditions associated with increased intestinal permeability. The concept of a "leaky gut" is transitioning from a fringe theory to a recognized physiological state with significant implications for public health, offering a promising new direction in the pursuit of optimal well-being.
The information presented in this article is intended for general informational purposes only and does not constitute medical advice. Always consult with a qualified healthcare professional for any health concerns or before making any decisions related to your health or treatment.