The landscape of modern medicine, particularly in surgical interventions, is being re-examined through the lens of rigorous scientific inquiry, with sham surgery trials revealing unsettling truths about some of the most widely adopted procedures. These studies, designed to isolate the true efficacy of surgical interventions from the powerful placebo effect, have cast a shadow of doubt on the effectiveness and safety of certain popular surgeries, raising critical questions about patient care and industry influence.
The Rise and Fall, and Resurgence of Intragastric Balloons
Intragastric balloons, first introduced with considerable enthusiasm in the 1980s, promised a less invasive approach to weight management. The concept was straightforward: a device implanted into the stomach and inflated to occupy a significant portion of its volume, thereby inducing satiety and reducing food intake. However, the history of medical devices is often marked by a premature entry into the market, driven by innovation rather than comprehensive evidence of efficacy and safety. Intragastric balloons were no exception.
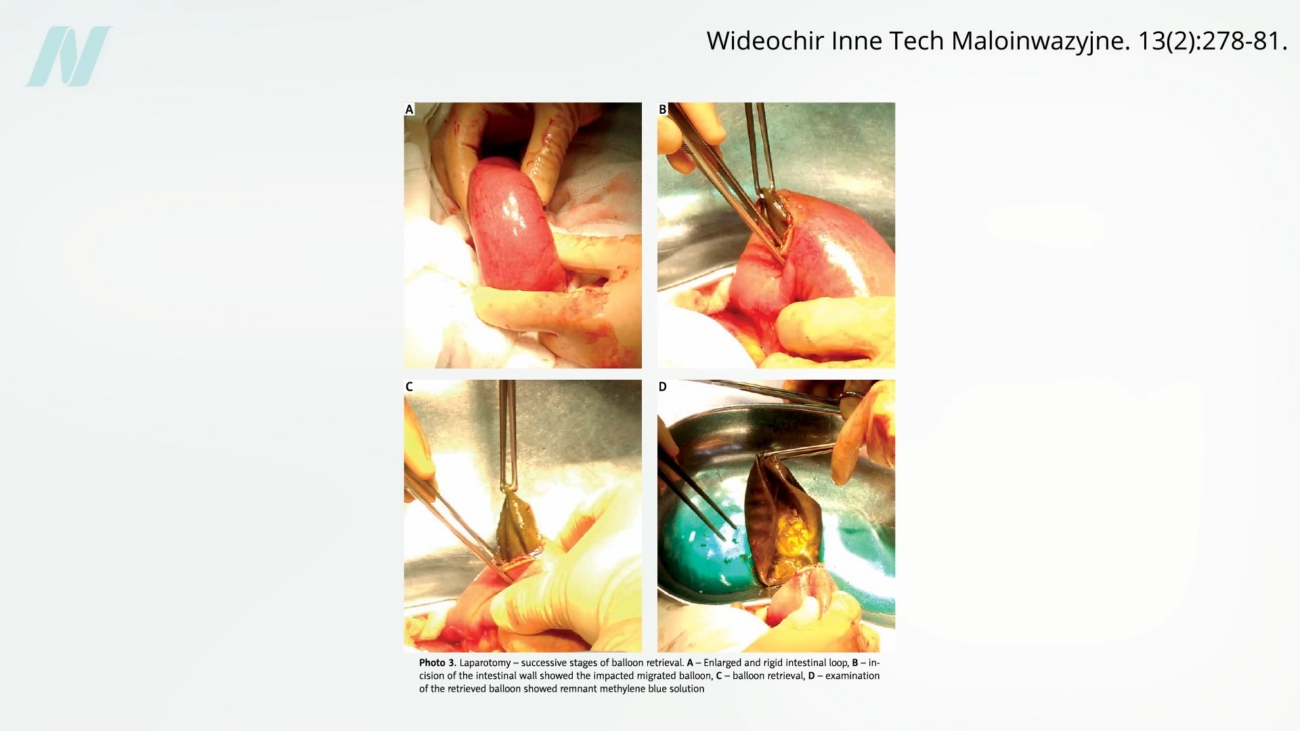
Early trials and clinical observations quickly exposed the limitations and potential dangers of these devices. A pivotal study conducted at the Mayo Clinic, for instance, found a startling failure rate: 8 out of 10 intragastric balloons spontaneously deflated. This mechanical failure posed a significant risk, as deflated balloons could migrate into the intestines, leading to potentially life-threatening bowel obstructions. Visual evidence, including images and video documentation, highlighted the severe consequences of such complications, underscoring the inherent risks associated with these devices.
Beyond the immediate risk of obstruction, the balloons also contributed to significant gastrointestinal distress. Even before deflation, studies indicated that intragastric balloons caused gastric erosions in approximately half of the patients, leading to damage of the stomach lining. The most concerning finding, however, was their questionable efficacy in promoting sustainable weight loss. When compared to simpler, non-surgical interventions like dietary changes and behavioral modification strategies, the weight loss achieved with intragastric balloons was often found to be negligible or temporary. Consequently, intragastric balloons were largely withdrawn from the market, their initial promise overshadowed by a clear lack of proven benefit and significant risks.
Despite this history, intragastric balloons have made a comeback. Following a hiatus of over three decades, the U.S. Food and Drug Administration (FDA) began approving a new generation of these devices in 2015. This resurgence coincided with the passage of the Sunshine Act, legislation aimed at increasing transparency in financial relationships between healthcare providers and the pharmaceutical and medical device industries. The Act mandates the disclosure of payments made to physicians by these entities, shedding light on potential conflicts of interest.
While public awareness of the financial ties between doctors and "Big Pharma" has grown, the influence of medical device companies on surgical practices may be less understood. Reports reveal that a substantial number of top physicians receive significant financial compensation from device manufacturers. For example, data from a single year indicated that the top 100 physician recipients of industry payments garnered an astonishing $12 million from device companies. Alarmingly, a significant minority of these physicians failed to disclose these financial relationships when publishing research, raising concerns about the objectivity of their findings and the potential for biased recommendations.

The Hidden Dangers of Reversible Procedures
While intragastric balloons are lauded for being reversible, a characteristic that distinguishes them from more invasive bariatric surgeries, this reversibility does not equate to safety. The FDA has issued numerous advisories detailing the serious risks associated with these devices, including the possibility of death. The mechanisms by which such fatalities can occur are often indirect but nonetheless severe. Persistent nausea and vomiting, common side effects reported by a majority of patients, can lead to severe complications. In extreme cases, forceful vomiting can cause a stomach perforation, a catastrophic event with a high mortality rate. These persistent gastrointestinal issues can also contribute to life-threatening nutrient deficiencies, further complicating patient recovery and well-being.
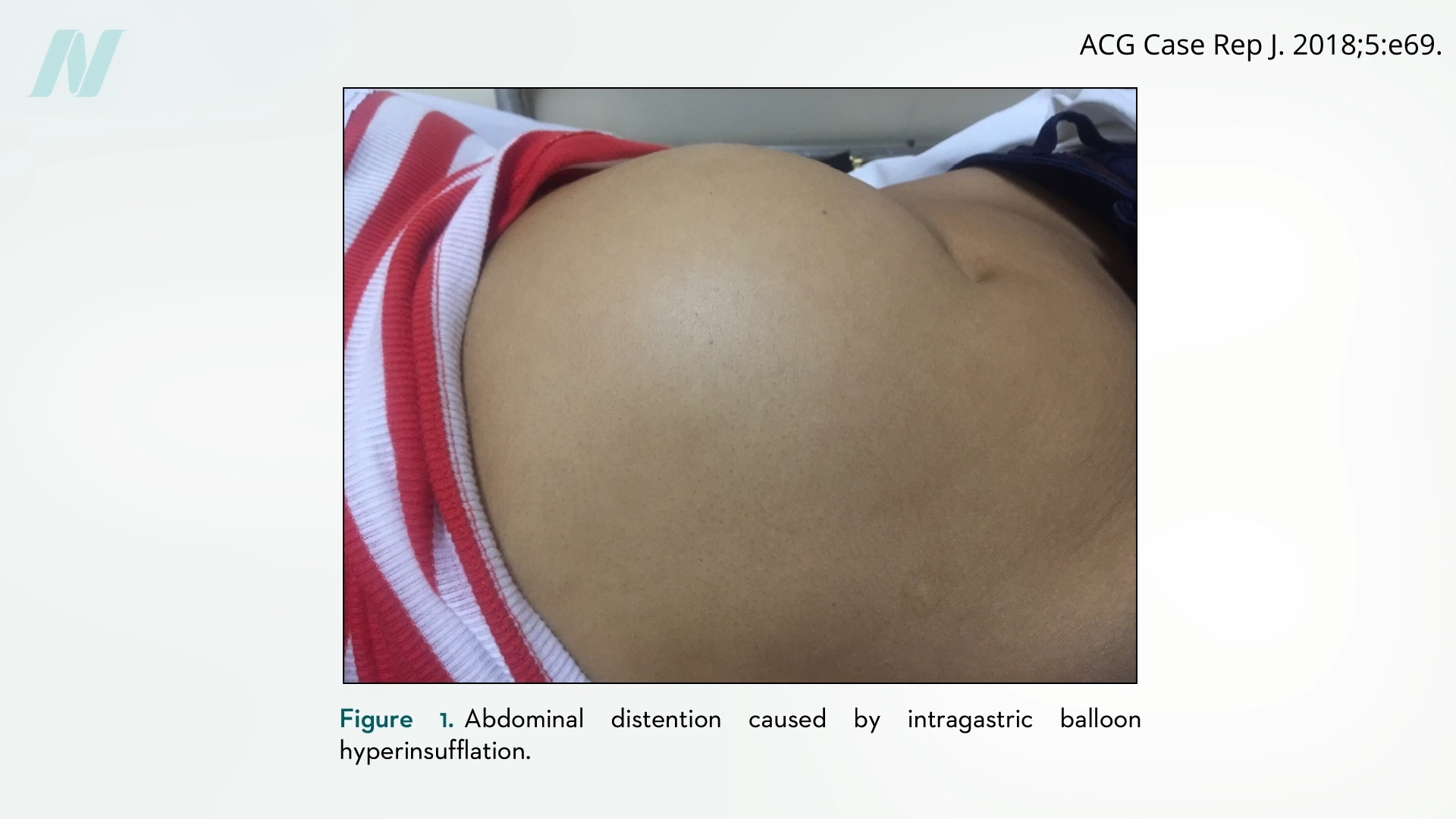
Complications arising from intragastric balloons are not limited to spontaneous deflation. A paradoxical issue has also emerged: sudden overinflation of the balloons. This phenomenon, observed in some cases, can lead to intense pain, severe vomiting, and abdominal distension, requiring urgent medical intervention. The unpredictability of these events adds another layer of concern to the use of these devices.
Interestingly, the phenomenon of spontaneous overinflation in medical devices is not entirely novel. Similar occurrences have been documented with breast implants, where implants have been observed to increase in volume inexplicably, sometimes by over 50%. The precise mechanisms behind this "spontaneously autoinflating" phenomenon remain underreported and poorly understood. It is noteworthy that some of the earliest experimental intragastric balloons were, in fact, adapted from breast implant technology, suggesting a shared underlying issue in device design or material behavior.
The Undermining Power of Sham Surgery Trials
The fundamental challenge in evaluating the effectiveness of any medical intervention, particularly surgery, lies in distinguishing the genuine therapeutic effect from the placebo effect. Industry-funded trials often present favorable outcomes, citing "notable weight loss." However, it is frequently difficult to isolate the specific contribution of the device or surgical technique from the accompanying "supervised diet and lifestyle changes" that are integral to these studies.
To address this methodological hurdle, researchers have employed the controversial yet scientifically crucial technique of sham surgery. In drug trials, this is achieved through the use of placebos – inert substances that mimic the active medication. However, in surgical research, the equivalent is a sham surgery, where a procedure is performed on a control group without any actual therapeutic intervention. This involves making incisions, mimicking surgical steps, and sometimes even using saline to simulate the splashing of fluid, all without operating on the target tissue.
The Knee Arthroscopy Revelation
A landmark study published in The New England Journal of Medicine in 2002 brought the concept of sham surgery into sharp focus and sparked considerable debate within the medical community. The study targeted arthroscopic surgery of the knee, the most commonly performed orthopedic procedure, which involves inserting a small camera and instruments into the knee joint to diagnose and treat various conditions, including osteoarthritis and injuries. Billions of dollars are invested annually in these procedures, with the assumption of significant patient benefit.
The study’s design was ethically contentious: patients suffering from knee pain were randomly assigned to either undergo the actual arthroscopic surgery or a sham procedure. In the sham group, surgeons made incisions and performed simulated surgical steps, including flushing the joint with saline, but did not cut or remove any tissue. The ensuing controversy was immense, with professional medical associations questioning the ethics of subjecting patients to unnecessary surgical exposure and the sanity of those who consented to such trials.
The results, however, were revelatory. While patients who received the actual surgery experienced improvement in their knee pain, a statistically similar degree of improvement was observed in the group that underwent the sham procedure. This finding suggested that the perceived benefits of arthroscopic knee surgery might be largely attributable to the placebo effect, the natural healing process, or other non-specific factors, rather than the surgical intervention itself. This study, along with subsequent research, has led to a significant reevaluation of the indications for and efficacy of arthroscopic knee surgery, with some guidelines now recommending non-operative treatments as the first line of defense for many knee conditions.
A Crisis of Confidence in Orthopedic Surgery
The implications of the knee arthroscopy trial extended beyond that single procedure. It initiated a broader crisis of confidence in orthopedic surgery, prompting similar investigations into other common interventions. Rotator cuff shoulder surgery, another highly prevalent procedure, is currently facing similar scrutiny. Research is increasingly exploring the effectiveness of these surgeries against placebo or conservative management, with preliminary findings echoing the earlier revelations.
The Intragastric Balloon Revisited: A Sham-Controlled Perspective
When intragastric balloons were subjected to sham-controlled trials, the results mirrored the concerning findings from orthopedic surgery. These rigorous studies demonstrated that both older and newer iterations of these devices often failed to provide any statistically significant weight-loss benefit compared to a sham procedure. Even in instances where some weight loss was observed, it was frequently temporary. This is partly due to the limited duration for which balloons are typically allowed to remain in place – usually around six months. Beyond this timeframe, the risk of deflation and subsequent complications increases significantly.
Attempts to prolong the use of balloons or to use them sequentially have also proven unsuccessful in achieving sustained long-term weight loss outcomes. Sham-controlled trials have indicated that any initial effects on appetite and satiety can diminish over time, as the body adapts to the presence of the device. This suggests that the perceived benefits of intragastric balloons may be largely illusory, driven by the psychological impact of undergoing a medical procedure rather than a direct physiological mechanism for sustainable weight management.
The Broader Implications: "Fake News" in Medicine?
The findings from sham surgery trials raise profound questions about the integrity of medical research and the dissemination of medical information. Doctors often pride themselves on their adherence to scientific principles, and the medical community rightly condemns the spread of misinformation, such as the anti-vaccination movement, where individuals "choose their own facts." However, the evidence emerging from sham surgery studies suggests that the medical field itself is not immune to its own forms of "fake news" and "alternative facts."
The fact that some popular surgeries, including those with significant financial backing from device manufacturers, have been shown to be ineffective, and in some cases, even detrimental (e.g., increasing the risk of progression to more invasive procedures like total knee replacement), is deeply troubling. It highlights the critical need for independent, rigorous research that prioritizes patient well-being over commercial interests.
The implications extend to how medical information is communicated to the public. Patients often rely on their physicians for unbiased advice, and when those physicians have financial ties to the devices or procedures they recommend, the potential for compromised decision-making is substantial. The Sunshine Act has been a positive step toward transparency, but ongoing vigilance and a commitment to evidence-based medicine are paramount.
The Path Forward: Towards Evidence-Based Patient Care
The lessons learned from sham surgery trials are invaluable. They underscore the importance of:
- Independent Research: Prioritizing studies funded by unbiased sources, free from the influence of device manufacturers and pharmaceutical companies.
- Rigorous Methodologies: Employing robust research designs, including sham-controlled trials, to accurately assess the efficacy and safety of medical interventions.
- Transparency and Disclosure: Ensuring full disclosure of all financial relationships between healthcare providers and industry to enable informed decision-making by patients and healthcare professionals alike.
- Patient Education: Empowering patients with accurate and comprehensive information about the risks and benefits of various treatment options, enabling them to participate actively in their healthcare decisions.
- Continuous Re-evaluation: Regularly reviewing and re-evaluating the effectiveness and safety of established medical procedures in light of new scientific evidence.
The ongoing exploration of these critical issues, as exemplified by the examination of "Extreme Weight-Loss Devices" and broader discussions on bariatric surgery, is essential for safeguarding patient health and maintaining public trust in the medical profession. The pursuit of sustainable weight loss, as detailed in comprehensive guides like "How Not to Diet," further emphasizes the need for evidence-based approaches that prioritize long-term health and well-being over quick fixes with unproven benefits and potentially harmful side effects. The medical community must confront these challenging revelations with a commitment to scientific integrity and an unwavering focus on the best interests of patients.