The global food supply chain, a complex network designed to nourish billions, is increasingly facing scrutiny over the presence of naturally occurring toxins that can pose significant health risks. Among these, ochratoxin, a mycotoxin produced by certain molds, has emerged as a subject of scientific and public health discussion. While regulatory bodies and industry stakeholders assert that current levels in commonly consumed foods are safe, a deeper dive into the scientific literature reveals a nuanced picture, highlighting the potential long-term implications for human health and the robust scientific methods employed to assess and mitigate these risks.
Understanding Ochratoxin: A Silent Contaminant
Ochratoxin is a potent nephrotoxin and a suspected human carcinogen. Its presence has been documented in a wide array of agricultural commodities, including cereals, coffee, dried fruits, wine, and pork. The contamination typically occurs during storage or processing when environmental conditions are conducive to mold growth. The classification of ochratoxin as a potential human carcinogen is primarily based on extensive animal studies. These studies have demonstrated its ability to induce kidney damage, immunotoxicity, and various forms of cancer in laboratory animals. However, translating these findings directly to human health outcomes has presented challenges, leading to ongoing scientific debate and the establishment of stringent regulatory guidelines.
Navigating the Risk Assessment Landscape
The process of determining acceptable daily intake (ADI) levels for substances like ochratoxin is a rigorous scientific undertaking. Regulatory agencies, such as the European Food Safety Authority (EFSA) and the U.S. Food and Drug Administration (FDA), rely on comprehensive toxicological data. A key methodology involves identifying a "benchmark dose" in animal models, which is the dose that elicits a 10% increase in a particular adverse effect, such as kidney pathology. To ensure a substantial margin of safety for the human population, this benchmark dose is then divided by a safety factor, often 500. This factor accounts for interspecies differences in susceptibility and variations within the human population.

For carcinogenic risks, a different approach is employed. Scientists identify the "tumor dose," the level at which a 5% increase in tumor incidence is observed in laboratory animals. From this, they extrapolate to determine the "negligible cancer risk intake." This calculation typically incorporates an even larger safety factor, often around 5,000-fold. This substantial safety margin is intended to ensure that even individuals with high dietary exposure to ochratoxin face a minimal, statistically insignificant risk of developing cancer over their lifetime.
Industry Assurances and Consumer Concerns
Major agricultural and food industry associations have consistently maintained that the ochratoxin levels found in the food supply, even in products frequently consumed by vulnerable populations like children, are well within safe limits. They often cite the robust safety factors employed in risk assessments to reassure consumers. For instance, in the context of oat-based cereals, industry representatives have stated that even for children with high consumption patterns, the lifetime cancer risk associated with ochratoxin exposure is considered negligible.
However, the basis for these assurances, particularly the extrapolation from animal studies and the application of large safety factors, has sometimes been questioned by independent researchers and public health advocates. The concern is that while these methods aim for prudence, they may not fully capture the complexities of human exposure and potential synergistic effects with other dietary or environmental factors. The question of how these "negligible risk" thresholds are derived and the underlying assumptions frequently arise in discussions about food safety regulations.
Scientific Evidence: The Double-Edged Sword of Whole Grains
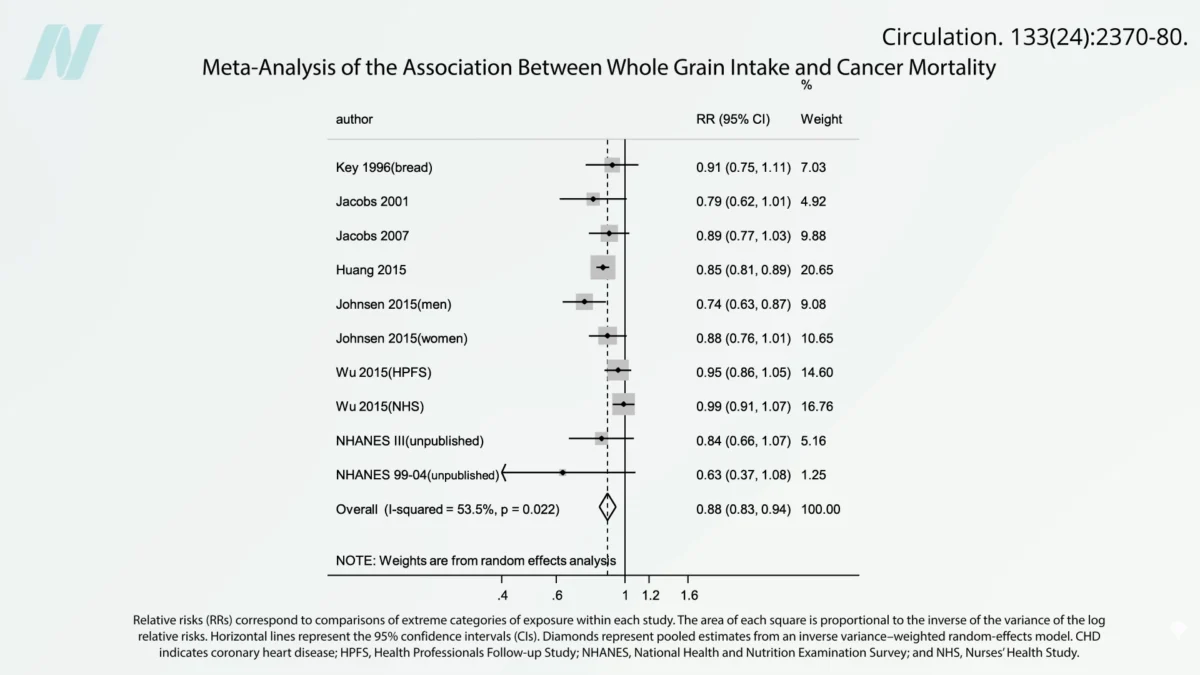
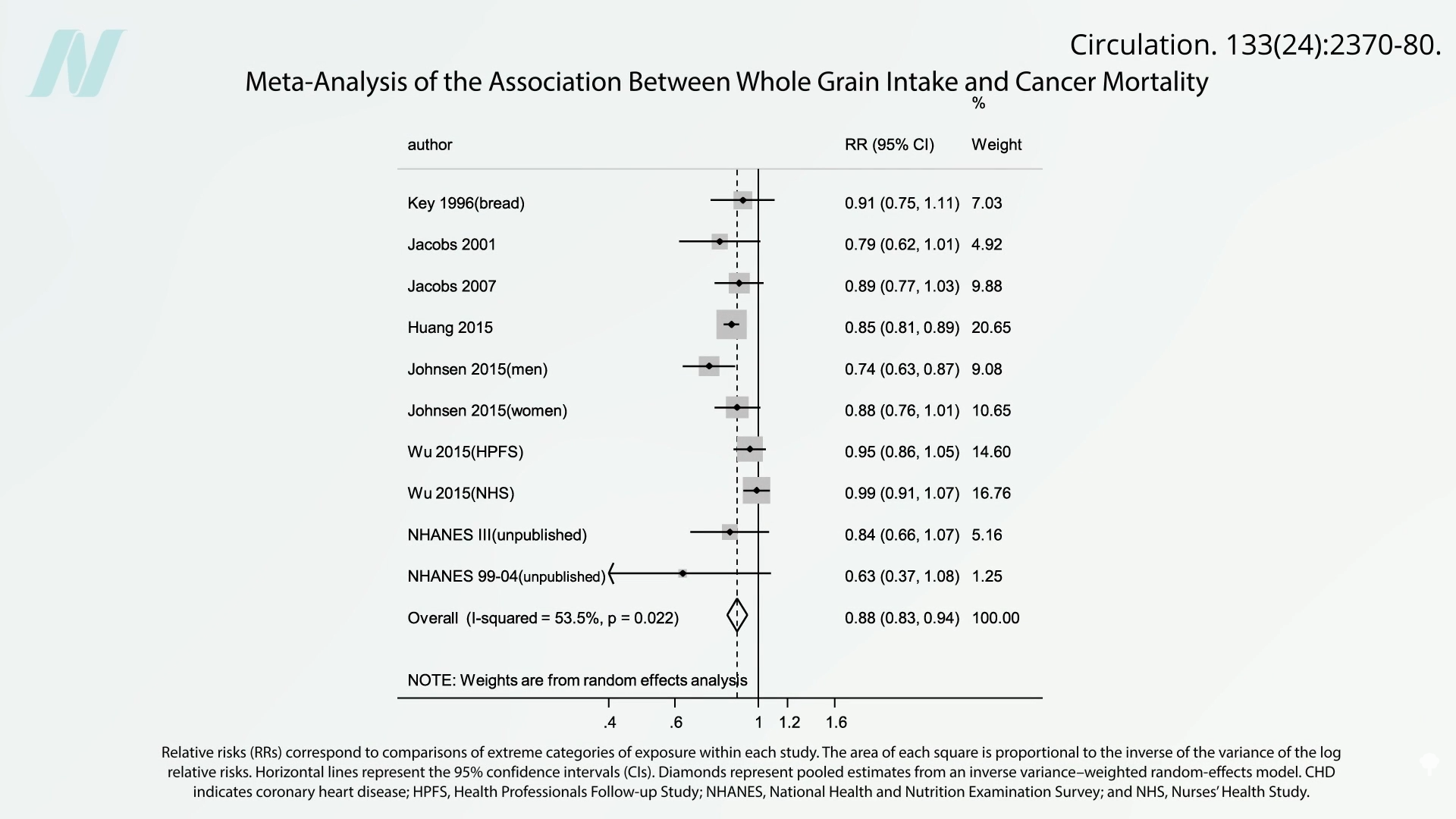
The debate surrounding ochratoxin takes on particular significance when considering the consumption of whole grains, a cornerstone of healthy dietary recommendations. Numerous epidemiological studies have established a strong inverse correlation between whole grain intake and mortality from various causes, including cardiovascular disease and cancer. For example, research published in leading nutritional journals has indicated that for every additional ounce of whole grains consumed daily, there is a statistically significant reduction in cancer mortality and all-cause mortality. These findings are robust, consistently observed across large-scale, long-term studies.

This observed protective effect of whole grains presents a fascinating paradox when juxtaposed with the presence of ochratoxin, a known contaminant. The healthy constituents of whole grains, such as their rich array of antioxidants, fiber, and phytochemicals, are believed to exert protective effects at the cellular level. These beneficial compounds may actively counteract the damage caused by mycotoxins, potentially by neutralizing free radicals and supporting cellular repair mechanisms. This suggests that the overall health benefits derived from consuming whole grains may significantly outweigh the potential risks posed by low-level ochratoxin contamination.
Broader Implications: Beyond Cereals
The impact of ochratoxin extends beyond breakfast cereals. Studies have detected its presence in other food categories, with varying prevalence and levels. Wine, for instance, has been identified as a potential source of ochratoxin exposure, with contamination rates and levels dependent on grape varietals, fermentation processes, and storage conditions. Similarly, pork products have also shown significant contamination. A notable percentage of tested pork samples have been found to contain ochratoxin, raising concerns for populations with high pork consumption.
The implications of these findings are multifaceted. For less healthful foods, the presence of ochratoxin can exacerbate their already negative health profile. In contrast, for inherently healthy foods like whole grains, the contaminant may slightly diminish their optimal benefits, but not to the extent of negating their profound positive impact on public health. This underscores the importance of a holistic dietary approach, where the overall quality and nutritional density of food choices play a crucial role in mitigating the risks associated with dietary contaminants.
A Chronology of Concern and Regulatory Response
The scientific understanding of ochratoxin’s toxicity has evolved over several decades. Early research in the 1960s and 1970s focused on its nephrotoxic effects in animals. By the 1980s and 1990s, its potential carcinogenic properties began to be investigated more thoroughly, leading to its classification as a possible human carcinogen by international health organizations. This scientific progress spurred the development of regulatory frameworks aimed at controlling ochratoxin levels in various food commodities.
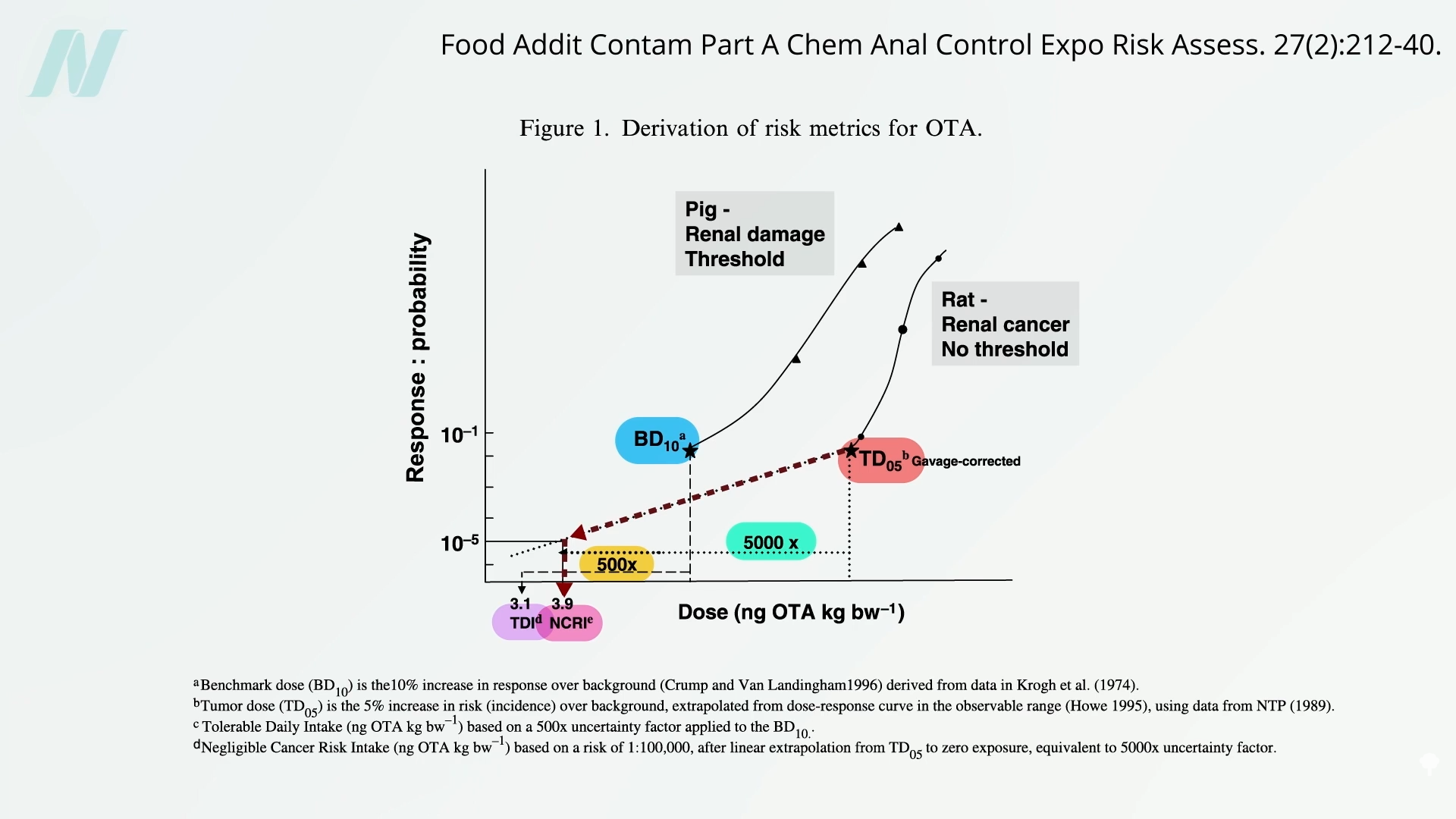
The establishment of maximum residue limits (MRLs) for ochratoxin in cereals, dried fruits, and other products began in the late 1990s and early 2000s across different global regions. These limits are periodically reviewed and updated as new scientific data emerges. For example, the European Union has specific regulations for ochratoxin A in foodstuffs, which are regularly assessed by EFSA. In the United States, the FDA monitors for mycotoxins, including ochratoxin, and takes action when levels exceed established guidance or pose an imminent health risk.
The ongoing scientific dialogue reflects a dynamic process of risk assessment and management. While regulatory bodies aim to set science-based standards, continuous research is essential to refine our understanding of ochratoxin’s effects, particularly in the context of modern dietary patterns and potential interactions with other food components.
Analysis and Future Outlook
The presence of ochratoxin in the food supply serves as a reminder of the inherent challenges in ensuring complete food safety. While the current regulatory approach, characterized by the use of extensive safety factors, aims to protect public health, the scientific community continues to explore more sophisticated methods for assessing cumulative and synergistic toxicological effects.
The findings regarding whole grains are particularly encouraging, suggesting that promoting a diet rich in nutrient-dense, plant-based foods is a powerful strategy for both disease prevention and mitigating the impact of unavoidable dietary contaminants. The research highlights that the beneficial compounds within these foods can offer a degree of protection against toxins like ochratoxin.
As scientific understanding advances, there is a growing emphasis on a "food systems" approach to safety, which considers the entire lifecycle of food, from farm to fork. This includes improving agricultural practices to minimize mold growth, enhancing post-harvest handling and storage, and developing more sensitive and cost-effective methods for detecting mycotoxins.
Ultimately, the narrative surrounding ochratoxin is not one of outright alarm, but rather of informed vigilance. It underscores the critical role of ongoing scientific research, robust regulatory oversight, and consumer education in navigating the complexities of our global food supply and making informed dietary choices that prioritize long-term health and well-being. The continued collaboration between scientists, policymakers, and the food industry will be paramount in addressing the challenges posed by mycotoxins and ensuring the safety and nutritional integrity of the foods we consume.