For centuries, the profound connection between our gut and overall health has been a subject of fascination and scientific inquiry. Hippocrates, the ancient Greek physician, famously declared, "All disease begins in the gut," a sentiment that resonates deeply with modern understanding of the microbiome. Our trillions of gut bacteria, collectively known as the microbiome, play a crucial role in numerous bodily functions, including digestion, nutrient absorption, and immune system regulation. When nourished with a diet rich in whole plant foods, these microbial allies thrive, producing beneficial compounds like butyrate, a short-chain fatty acid vital for gut health. Conversely, a diet high in animal products, such as cheese, eggs, seafood, and meat, can foster the growth of bacteria that produce detrimental compounds, most notably trimethylamine N-oxide (TMAO).
TMAO has emerged as a significant marker of cardiovascular risk, linked to heart disease and stroke. However, recent research has expanded our understanding of its far-reaching impact, revealing associations with a spectrum of serious health conditions. Beyond its well-established role in cardiovascular ailments, TMAO has been implicated in inflammatory diseases like psoriatic arthritis and metabolic disorders such as polycystic ovary syndrome. The growing body of evidence suggests that TMAO’s influence extends to some of the leading causes of mortality in developed nations, prompting a deeper investigation into its mechanisms and potential interventions.
TMAO: A Widespread Culprit in Chronic Disease
The implications of elevated TMAO levels are particularly concerning when considering the top ten causes of death in the United States. While its link to heart disease (the leading killer) and stroke (the fifth leading killer) is well-documented, recent studies have uncovered a concerning association between TMAO blood levels and the increased risk of various cancers, which collectively represent the second leading cause of death. The precise mechanisms by which TMAO contributes to cancer development are still under investigation, but potential pathways include promoting inflammation, inducing oxidative stress, causing DNA damage, and disrupting protein folding—all processes that can contribute to oncogenesis.
The impact of TMAO is not limited to cardiovascular and oncological concerns. Chronic obstructive pulmonary disease (COPD), the fourth leading killer, also shows a connection. TMAO has been associated with premature mortality in patients experiencing exacerbated COPD, though it is suspected that these outcomes may be indirectly linked to cardiovascular complications that often accompany severe respiratory disease.
The link between TMAO and stroke is particularly direct and intuitive. Higher TMAO levels are associated with elevated blood pressure, a major risk factor for stroke. Furthermore, individuals with increased TMAO are more prone to blood clot formation, especially those with atrial fibrillation, a condition that significantly increases stroke risk. Studies indicate that individuals with higher TMAO levels not only face a greater likelihood of experiencing a stroke but also tend to suffer more severe outcomes and a fourfold increase in the odds of death following such an event.
Alzheimer’s disease, the sixth leading cause of death, also falls under TMAO’s potential influence. TMAO has been detected in human cerebrospinal fluid, the protective liquid surrounding the brain, and elevated levels have been observed in individuals with mild cognitive impairment and Alzheimer’s disease dementia. Research suggests that within the brain, TMAO can induce neuronal senescence (age-related cellular deterioration), exacerbate oxidative stress, impair mitochondrial function, and interfere with mTOR signaling pathways. These cellular and molecular disruptions collectively contribute to brain aging and cognitive decline.
The list of TMAO-associated conditions continues. Diabetes, the seventh leading killer, shows a significant correlation, with individuals exhibiting higher TMAO levels being approximately 50% more likely to develop the disease. Pneumonia, the eighth leading killer, also demonstrates a link, where TMAO levels can predict fatal outcomes in patients, even in the absence of apparent heart disease. Kidney disease, the ninth leading cause of death, is strongly related to TMAO. Elevated TMAO levels are indicative of impaired kidney function and predict poorer prognoses. In a longitudinal study, over half of chronic kidney disease patients with average or higher TMAO levels at the study’s outset died within five years, starkly contrasting with the nearly 90% survival rate among those in the lowest third of TMAO levels.
The Search for Solutions: Dietary Intervention and Beyond
Given TMAO’s pervasive impact on health, understanding how to mitigate its levels in the body is paramount. Since TMAO originates from dietary precursors, a logical approach involves limiting the intake of foods rich in choline and carnitine, compounds that gut bacteria convert into trimethylamine (TMA), which is then oxidized to TMAO. However, these nutrients are widely distributed in common food sources, particularly animal products like meat, eggs, and dairy. This widespread presence makes strict dietary restriction of TMA-containing nutrients a challenging, and for many, impractical endeavor.
This dietary challenge has spurred interest in alternative interventions, including exploring the potential of modifying the gut microbiome itself. The question arises: if the microbiome of individuals following plant-based diets inherently protects against the toxic effects of TMAO, could altering one’s gut flora through other means offer a similar benefit? This line of inquiry has led to investigations into the efficacy of fecal microbiota transplantation (FMT), a procedure where the gut bacteria from a healthy donor are introduced into a recipient’s digestive system.
Fecal Transplants: A Potential Shortcut?
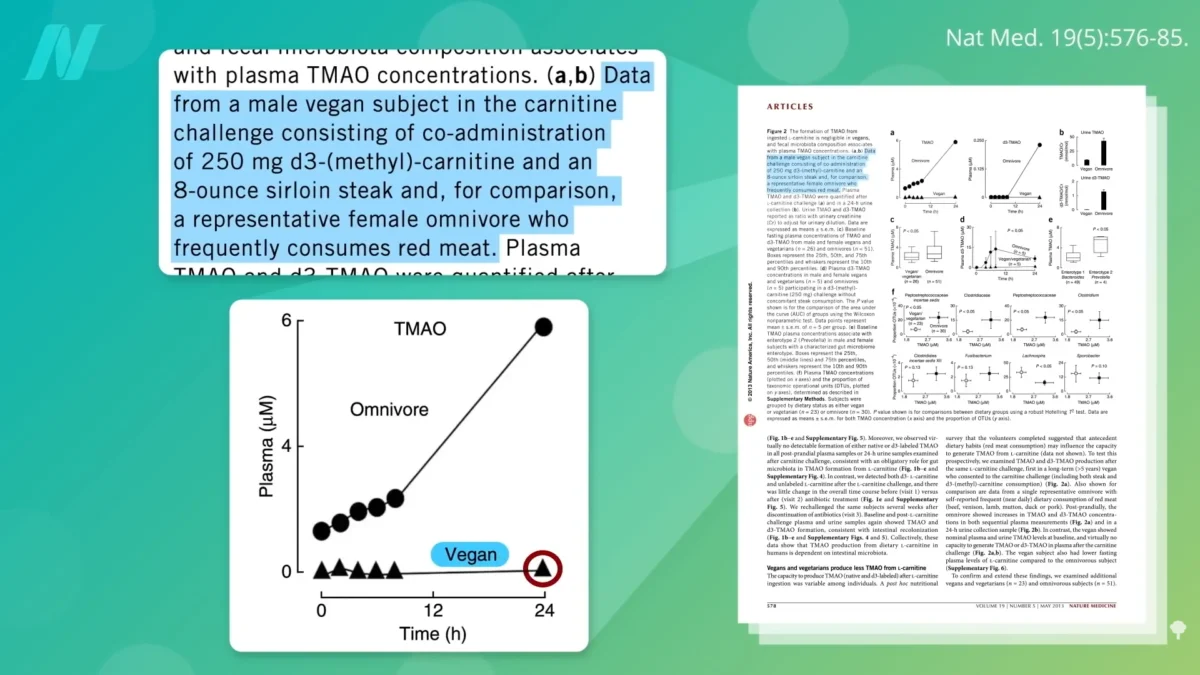
The concept of "swapping gut flora" through fecal transplantation, particularly from donors with a protective microbiome, has garnered attention. The initial hypothesis centered on the observation that individuals who strictly adhere to a plant-based diet produce significantly less TMAO, even when consuming carnitine-rich foods like steak. This suggests that their gut microbiome has evolved a different metabolic profile, less conducive to TMA production. For instance, studies have shown that vegans, after being on a plant-based diet for at least a year, produce negligible amounts of TMAO when given a steak, a stark contrast to meat-eaters who exhibit a substantial increase. This difference is attributed to the absence of "steak-eating bugs" in the vegan gut, implying that the microbiome composition is key.

Intrigued by this, researchers explored whether a fecal transplant from vegan donors could effectively lower TMAO levels in recipients. A notable study involved a double-blind, randomized, controlled trial where participants received either vegan fecal matter or their own microbiota via a nasogastric tube. The results, however, were not as promising as initially hoped. The intervention failed to significantly improve TMAO levels in the recipient group.
Several factors may have contributed to these findings. Firstly, the vegans recruited for this specific study were not required to have maintained their plant-based diet for an extended period, unlike in earlier observational studies where long-term vegans demonstrated virtually no TMAO production. This suggests that the baseline microbiome differences might not have been as pronounced. Consequently, the transplanted fecal matter may have already possessed some capacity to produce TMAO.
Secondly, the study design, while robust in its methodology, presented inherent challenges. The researchers deliberately avoided switching participants to a plant-based diet post-transplant. Their rationale was to isolate the effect of the fecal transplant alone, as a dietary change independently influences the microbiome. However, this decision meant that recipients continued their omnivorous diets, which could have counteracted the potential benefits of the transplanted vegan microbiota. The researchers noted that the lack of improvement could be "related to limited baseline microbiome differences and continuation of an omnivorous diet" after the procedure.
The Enduring Importance of Diet
The findings from the fecal transplant study underscore a critical point: there may be no shortcuts to optimizing gut health and mitigating TMAO production. While the allure of a quick fix through microbial transplantation is understandable, the current evidence suggests that sustained dietary habits play a foundational role. The gut microbiome is a dynamic ecosystem, highly responsive to dietary inputs. Long-term adherence to a plant-based diet appears to cultivate a microbiome that is inherently resistant to TMAO formation, a resilience that may be difficult to replicate solely through a single intervention like fecal transplantation, especially without concomitant dietary changes.
The implications of these findings are significant for public health strategies aimed at reducing the burden of chronic diseases linked to TMAO. While research into microbiome manipulation continues, the most reliable and evidence-based approach to lowering TMAO levels and promoting overall well-being remains a commitment to a whole-foods, plant-based diet. This dietary pattern not only starves the TMAO-producing bacteria but also nourishes the beneficial microbes, fostering a healthier gut environment that can confer protection against a wide array of chronic illnesses.
Looking Ahead: Research and Individual Action
The scientific community continues to unravel the intricate relationship between diet, the microbiome, and human health. Ongoing research aims to further elucidate the specific bacterial species and metabolic pathways involved in TMAO production and degradation. Understanding these mechanisms could pave the way for more targeted interventions in the future, potentially including precisely engineered probiotics or prebiotics.
However, for individuals seeking to proactively manage their TMAO levels and reduce their risk of associated diseases, the message is clear: prioritize dietary changes. Embracing a diet rich in fruits, vegetables, legumes, and whole grains is not merely about limiting detrimental compounds but about actively cultivating a gut environment that supports health and longevity. The ancient wisdom of Hippocrates continues to hold true – the gut remains a central arena for health and disease, and nourishing it wisely is the most potent strategy available today.
Doctor’s Note:
For individuals interested in potentially contributing to fecal microbiota transplantation research, information on becoming a donor can be found through specific research initiatives and institutional guidelines. The process typically involves rigorous screening to ensure donor health and suitability.
Further exploration into the multifaceted role of TMAO in human health can be found in related scientific literature and public health resources. The microbiome topic page offers a comprehensive overview of the latest research in this rapidly evolving field.