The ancient wisdom of Hippocrates, who declared nearly 2,500 years ago that "All disease begins in the gut," is increasingly being validated by modern scientific research. The intricate ecosystem within our digestive tract, known as the microbiome, plays a pivotal role in our overall health, influencing everything from nutrient absorption to immune function and even mental well-being. Emerging research highlights a particularly concerning compound produced by gut bacteria: trimethylamine N-oxide (TMAO). While the microbiome of individuals following plant-based diets appears to offer protection against TMAO’s toxic effects, a compelling question arises: can we harness the power of gut flora by directly manipulating it, such as through fecal microbiota transplantation, to achieve similar protective benefits?
TMAO: A Growing Concern in Human Health
TMAO is a metabolic byproduct generated when gut bacteria break down certain dietary components, primarily choline and L-carnitine. These nutrients are abundant in animal-derived foods such as red meat, eggs, dairy products, and seafood. When consumed, these foods are metabolized by specific gut microbes, producing trimethylamine (TMA), which is then absorbed into the bloodstream and oxidized by the liver into TMAO.
Historically, TMAO was primarily associated with cardiovascular diseases. Studies have demonstrated a clear link between elevated TMAO levels and an increased risk of heart disease and stroke. The mechanisms underlying this association are multifaceted. TMAO can contribute to the progression of atherosclerosis by promoting cholesterol accumulation in artery walls and enhancing platelet reactivity, making blood clots more likely to form. Furthermore, TMAO has been implicated in raising blood pressure, a significant risk factor for both heart disease and stroke. Research has shown that individuals with higher TMAO levels not only experience more severe strokes but also face a fourfold increase in the odds of mortality following such an event.
However, the detrimental reach of TMAO extends far beyond cardiovascular health. More recent investigations have uncovered its association with a wider spectrum of diseases, painting a concerning picture of its systemic impact. Psoriatic arthritis, a chronic inflammatory condition, has been linked to elevated TMAO levels. Polycystic ovary syndrome (PCOS), a complex hormonal disorder affecting women of reproductive age, has also shown an association with higher TMAO concentrations.
Perhaps most alarmingly, TMAO is now being implicated in an increased risk of various cancers, which collectively represent the second leading cause of death in the United States. While the exact mechanisms are still under investigation, the inflammatory potential of TMAO is a leading hypothesis. Chronic inflammation is a known driver of cancer development, and TMAO may contribute through various pathways, including inducing oxidative stress, damaging DNA, and disrupting protein folding, all of which can foster a pro-cancerous cellular environment.
The impact of TMAO is also evident in other leading causes of mortality. For instance, it is associated with premature death in patients experiencing exacerbated Chronic Obstructive Pulmonary Disease (COPD), although this is suspected to be an indirect effect stemming from increased cardiovascular complications in these vulnerable individuals. Even neurological conditions are not spared. TMAO has been detected in human cerebrospinal fluid, the fluid that bathes the brain. Studies have revealed higher TMAO levels in individuals experiencing mild cognitive impairment and those diagnosed with Alzheimer’s disease dementia. Within the brain, TMAO has been shown to induce neuronal senescence (age-related deterioration of brain cells), exacerbate oxidative stress, impair mitochondrial function essential for cellular energy production, and interfere with crucial signaling pathways like mTOR, all of which contribute to brain aging and cognitive decline.
Diabetes, the seventh leading killer, also shows a connection. Individuals with higher TMAO levels are approximately 50% more likely to develop diabetes. Pneumonia, the eighth leading cause, sees TMAO as a predictor of fatal outcomes, even in patients without evident heart disease. Kidney disease, the ninth leading killer, demonstrates a strong relationship with TMAO. Elevated TMAO levels are significantly correlated with impaired kidney function and predict poorer outcomes, with a stark difference observed in survival rates: over a five-year period, more than half of chronic kidney disease patients with average or higher TMAO levels died, whereas nearly 90% of those in the lowest third of TMAO levels remained alive.
Dietary Interventions and the Microbiome: A Protective Alliance
The profound influence of the gut microbiome on TMAO production and its subsequent health implications naturally leads to the question of how to mitigate these risks. Given that TMAO originates from dietary precursors, a straightforward approach would be to limit the intake of foods rich in choline and L-carnitine, such as meat, eggs, and dairy. However, these nutrients are widespread in many common foods, making strict dietary restriction potentially impractical for many individuals. This limitation has spurred interest in alternative strategies, particularly those that leverage the power of the microbiome itself.
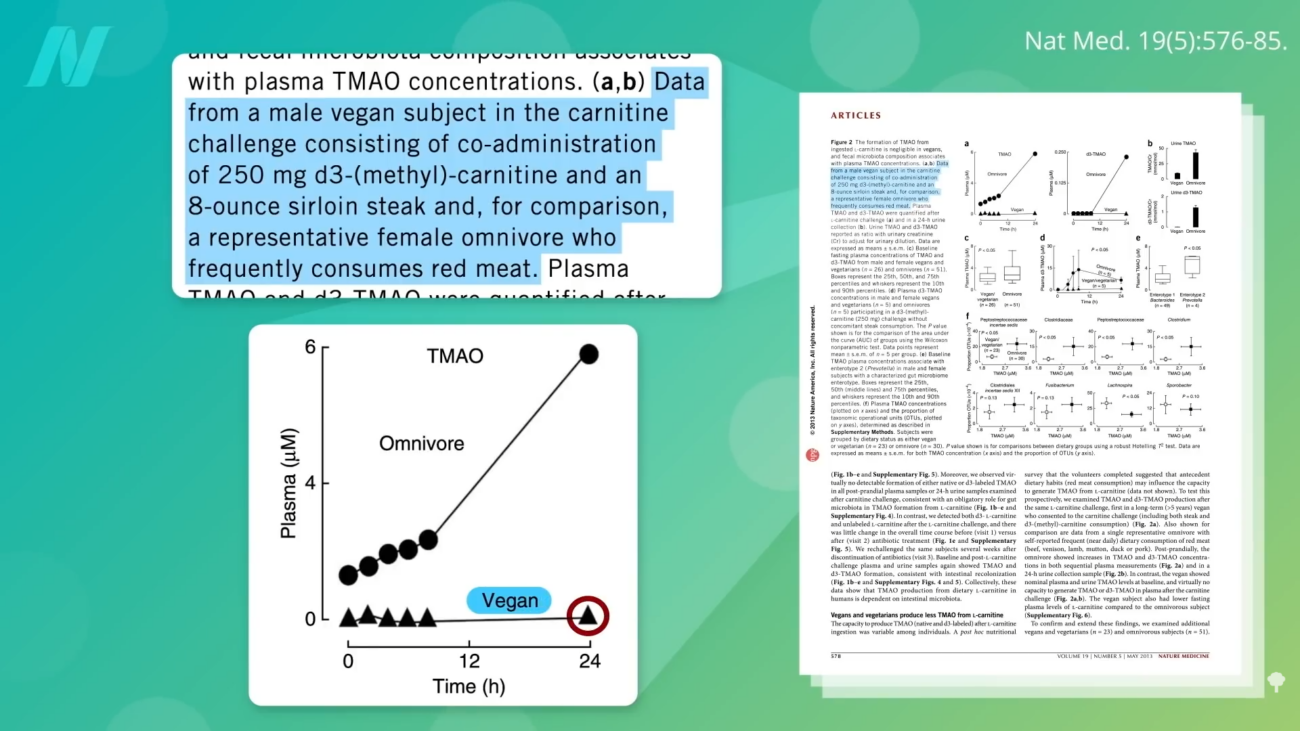
The observation that individuals adhering to plant-based diets exhibit a different gut microbial composition, one that appears to be protective against the toxic effects of TMAO, provides a crucial clue. It is hypothesized that the gut flora of plant-based eaters has not been "trained" to efficiently metabolize the TMAO precursors found abundantly in animal products. When a vegan consumes a meal rich in carnitine, for example, they produce significantly less TMAO compared to a meat-eater, even with the same dietary intake. This suggests that the microbial community, rather than the specific nutrient in isolation, plays a pivotal role in the metabolic outcome.

The question then becomes: can this protective microbial environment be transferred? This line of inquiry has led to the exploration of fecal microbiota transplantation (FMT), a medical procedure involving the transfer of fecal matter from a healthy donor to a recipient, with the aim of restoring a balanced and healthy gut microbiome. Specifically, researchers have investigated the potential of using fecal matter from individuals following a strict vegan diet – a "vegan fecal transplant" – as a means to reduce TMAO levels in recipients.
Early studies exploring this concept yielded intriguing, though not entirely conclusive, results. One notable investigation sought to assess the efficacy of vegan fecal transplants in lowering TMAO levels. In a double-blind, randomized, controlled trial, participants received either fecal matter from vegan donors or their own previously stored fecal matter, administered via a nasogastric tube. The primary outcome of this trial was to observe a reduction in TMAO levels post-transplantation.
However, the results of this particular study did not demonstrate the desired improvement. Several factors may have contributed to this outcome. Firstly, the vegan donors recruited for this study were found to be producing TMAO themselves, albeit at lower levels than omnivores. This contrasts with findings from other research where vegans exhibited virtually no TMAO production. The discrepancy might be attributed to variations in the duration of adherence to a vegan diet among the donor populations. In one study, vegans had maintained their diet for at least a year, potentially allowing for more profound shifts in their gut microbiome composition. In the transplanted study, the duration of veganism for donors was not specified, and the recipients did not exhibit significant changes in TMAO levels two weeks after the transplant. Furthermore, the vegan fecal matter itself appeared to possess some baseline capacity to produce TMAO.
The researchers posited that the failure to achieve significant improvement could be linked to limited baseline microbiome differences between donors and recipients, and crucially, the continuation of an omnivorous diet by the recipients after the transplant. The rationale behind not switching participants to a plant-based diet during the study was to isolate the effect of the fecal transplant. However, it is acknowledged that dietary changes alone can profoundly alter the microbiome. Without a concurrent shift in diet, the introduced microbial community may struggle to establish dominance or function optimally in the face of ongoing exposure to TMAO-promoting food sources.
This highlights a critical point: the effectiveness of any intervention aimed at modulating the gut microbiome, whether through diet or transplantation, is likely contingent upon the overall dietary context. If the goal is to foster a microbiome that protects against TMAO, then continuing to consume a diet rich in TMAO precursors may counteract the benefits of a microbial shift.
The Implications and Future Directions
The exploration of vegan fecal transplants, while not yielding immediate success in the tested protocol, underscores a significant scientific endeavor. It signifies a shift towards understanding and manipulating the microbiome as a therapeutic target for chronic diseases. The research suggests that simply introducing "vegan poop" may not be a shortcut to a healthier gut if the recipient’s lifestyle, particularly their diet, remains unchanged.
The underlying principle remains consistent: the gut microbiome is a dynamic entity shaped by dietary habits. A diet rich in whole plant foods, abundant in fiber, fuels beneficial bacteria that produce short-chain fatty acids like butyrate, which are vital for gut health. Conversely, a diet high in animal products provides substrates for bacteria that generate harmful compounds like TMAO.
The findings from these investigations point towards a more holistic approach. While direct microbiome interventions like FMT hold promise, they may be most effective when integrated with significant dietary modifications. The long-term adherence to a plant-based diet appears to be a powerful, albeit gradual, method for cultivating a gut microbiome that inherently resists the production of TMAO. This natural adaptation process, fostered by consistent dietary choices, suggests that there may be no easy shortcuts. The most reliable path to mitigating the risks associated with TMAO, and potentially improving overall health, lies in embracing a healthier diet.
For individuals interested in contributing to this field of research, opportunities to become fecal transplant donors exist. Information on how to become a "fecal transplant super donor" is available, offering a tangible way to participate in advancing our understanding of gut health and its therapeutic potential.
The ongoing research into TMAO and its multifaceted impact on human health is a testament to the evolving understanding of the gut microbiome’s central role. As scientific inquiry continues, further studies will likely refine our understanding of optimal microbiome modulation strategies, potentially integrating dietary interventions with more targeted microbial therapies to combat the growing burden of chronic diseases. The journey from Hippocrates’ ancient observation to the cutting edge of microbiome research underscores the enduring importance of our gut in the intricate tapestry of human health.