The practice of fasting, dating back over 2,400 years to the time of Hippocrates, is experiencing a resurgence in scientific inquiry, particularly within the realm of cancer treatment. Historically, physicians observed that the loss of appetite in the sick was not merely a symptom but often an inherent, albeit sometimes uncomfortable, aspect of the body’s defense mechanisms. This ancient wisdom is now being rigorously examined through modern scientific lenses, revealing a complex interplay between caloric restriction, immune function, and the fight against cancer.
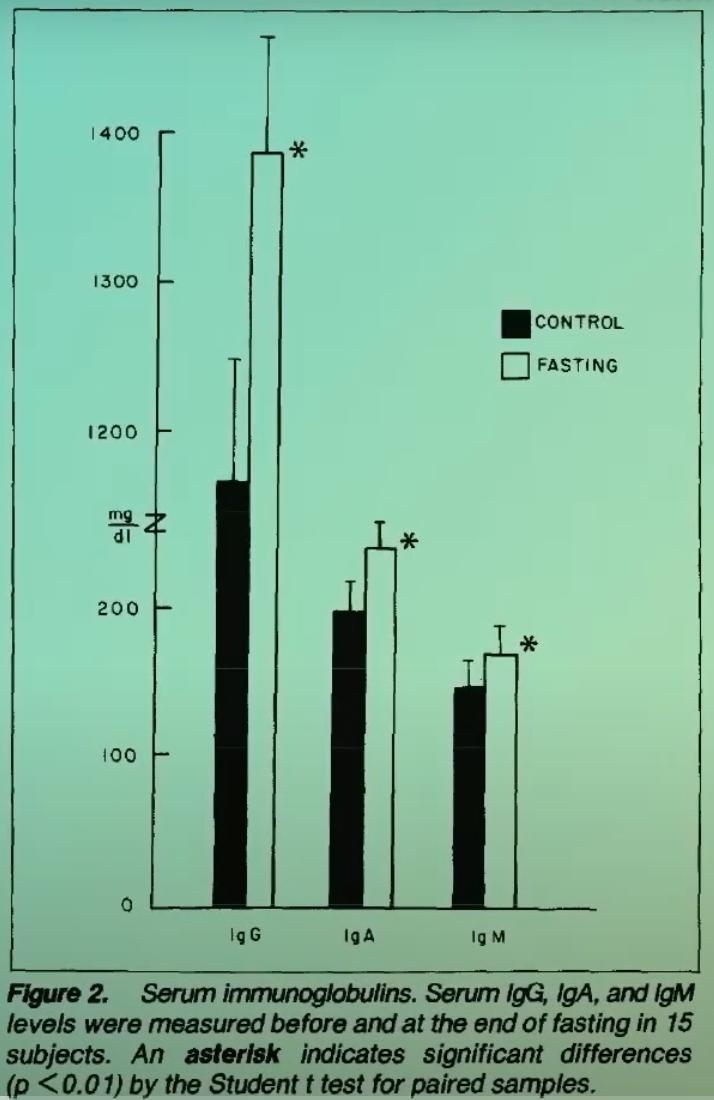
The intuitive link between illness and reduced food intake, alongside fever, has long been recognized as a cardinal sign of infection. While frequently viewed as an undesirable consequence of sickness, research now suggests that this decreased consumption is an active and potentially beneficial defense strategy. Although prolonged undernutrition can indeed compromise the immune system, short-term periods of reduced food intake appear to bolster immune capabilities. Early studies, such as those conducted on mice, provided compelling evidence: blood from starved rodents demonstrated an almost eightfold increase in its capacity to neutralize invading bacteria when tested in vitro. This dramatic enhancement was attributed to a significant boost in the pathogen-killing efficiency of their white blood cells. The critical question then became whether these findings translated to human physiology, especially in the context of cancer.
The Immune System’s Awakened Appetite for Cancer
The human immune system, a complex network of cells and processes, plays a pivotal role in identifying and eliminating threats, including cancerous cells. Natural killer (NK) cells, a critical component of this defense, are particularly adept at recognizing and destroying cells that display abnormal surface markers, a common characteristic of tumor cells. Research has explored how fasting might influence the activity of these crucial immune defenders.
In a notable study, human participants who underwent a two-week fasting regimen, consuming only 80 calories per day, exhibited remarkable immunological changes. Not only did their white blood cells demonstrate enhanced bacteria-killing abilities and increased antibody production, mirroring the rodent findings, but their natural killer cell activity saw an impressive average increase of 24%. This surge in NK cell function is particularly significant, as these cells are directly involved in the immunosurveillance against nascent tumors. To quantify this effect, researchers directly tested the participants’ NK cells against K562 cells, a human leukemia cell line, observing their enhanced cytotoxic potential. This suggests that fasting could potentially "stimulate the appetite of the immune system for cancer," a poetic yet scientifically grounded concept that highlights the potential for enhanced immune surveillance and response against malignancies.

Understanding Cancer Cachexia: A Complex Challenge
Despite these promising immunological findings, fasting has not historically been a mainstream component of cancer treatment. A primary reason for this lies in the pervasive challenge of cancer cachexia. This debilitating syndrome, characterized by involuntary weight loss and muscle wasting, is a significant contributor to morbidity and mortality in many cancer patients. Historically, a central tenet of palliative cancer care has been to maintain patient weight and combat this wasting effect, making the idea of intentionally restricting food intake seem counterintuitive, if not detrimental.
Tumors, by their very nature, are aggressive and rapidly growing entities that demand substantial energy and protein to fuel their expansion. This metabolic demand often leads to a systemic reprogramming of the body, where it begins to catabolize its own tissues to supply the tumor’s needs. This process is often exacerbated by inflammation, a widespread inflammatory response triggered by the presence of the tumor. The weight loss observed in cancer cachexia differs fundamentally from simple starvation; it is often refractory to nutritional repletion alone, indicating a more profound metabolic derangement orchestrated by the tumor. Consequently, therapeutic nutritional interventions aimed at reversing cachexia frequently prove insufficient. Therefore, the most effective strategy for managing cancer cachexia remains the direct treatment and eradication of the underlying cancer itself. The observation that forcing extra nutrition onto cancer patients might inadvertently fuel tumor growth, much like a fetus prioritizes nutrient acquisition during pregnancy, suggests that the natural loss of appetite experienced by some cancer patients might, paradoxically, be a protective mechanism.
The Limitations of Conventional Chemotherapy and the Emergence of Fasting as a Protective Strategy
For decades, chemotherapy has been a cornerstone of cancer treatment, employing drugs that primarily target rapidly dividing cells, including cancer cells, often through mechanisms involving DNA damage and free radical production. While initially believed to be highly selective for tumor cells, it became evident that these potent agents also inflict significant damage on healthy, rapidly dividing normal cells. This collateral damage leads to a cascade of dose-limiting side effects, including bone marrow suppression, immune system compromise, profound fatigue, gastrointestinal distress, and in severe cases, even death. Furthermore, the DNA damage inflicted on healthy cells can, ironically, increase the risk of secondary cancers later in life.
Attempts to mitigate these side effects through the use of cell-protective drugs have yielded mixed results. While such agents can reduce the toxicity, they may also inadvertently shield cancer cells, potentially diminishing the overall therapeutic efficacy of the chemotherapy. This limitation has spurred a search for alternative or adjunctive strategies that can enhance treatment effectiveness while preserving healthy tissues. Enter fasting.
The role of short-term fasting in cancer prevention and treatment is beginning to be recognized. Emerging research suggests that fasting protocols implemented before and immediately following chemotherapy sessions may not only significantly mitigate the harsh side effects but could also render cancer cells more susceptible to the treatment. This dual action is particularly compelling. During periods of caloric deprivation, healthy cells are believed to shift their metabolic state from active growth to a mode of maintenance and repair. In contrast, tumor cells, driven by their inherent growth-promoting mutations, often struggle to adapt to this scarcity, unable to downregulate their rapid proliferation. This inability to enter a quiescent state might represent a critical vulnerability, an "Achilles’ heel," for many types of cancer.
The differential response of healthy versus cancerous cells to short-term fasting theoretically allows chemotherapy to inflict greater DNA damage and induce cell death preferentially in tumor cells, while leaving healthy cells relatively unharmed. This protective effect of fasting could allow for higher chemotherapy doses to be administered safely, potentially increasing treatment efficacy. While this remains a compelling theory, its validation in human trials is ongoing.
Pre-clinical Evidence: Fasting’s Impact in Animal Models
Studies in animal models have provided significant insights into the potential of fasting in cancer treatment. In rodents, fasting alone has demonstrated efficacy comparable to conventional chemotherapy in certain contexts. Furthermore, the aggressive growth of tumors was observed to be significantly curtailed by radiation therapy. However, the combination of radiation therapy with alternate-day fasting yielded even more pronounced results, suggesting a synergistic effect. Intriguingly, alternate-day fasting alone appeared to achieve outcomes similar to radiation therapy in some of these preclinical models, underscoring the potent anti-tumor effects of caloric restriction. While these findings in mice and dogs are encouraging, the crucial step of translating these benefits to human cancer patients requires careful and extensive clinical investigation.
Human Trials: Navigating the Evidence and Patient Experiences
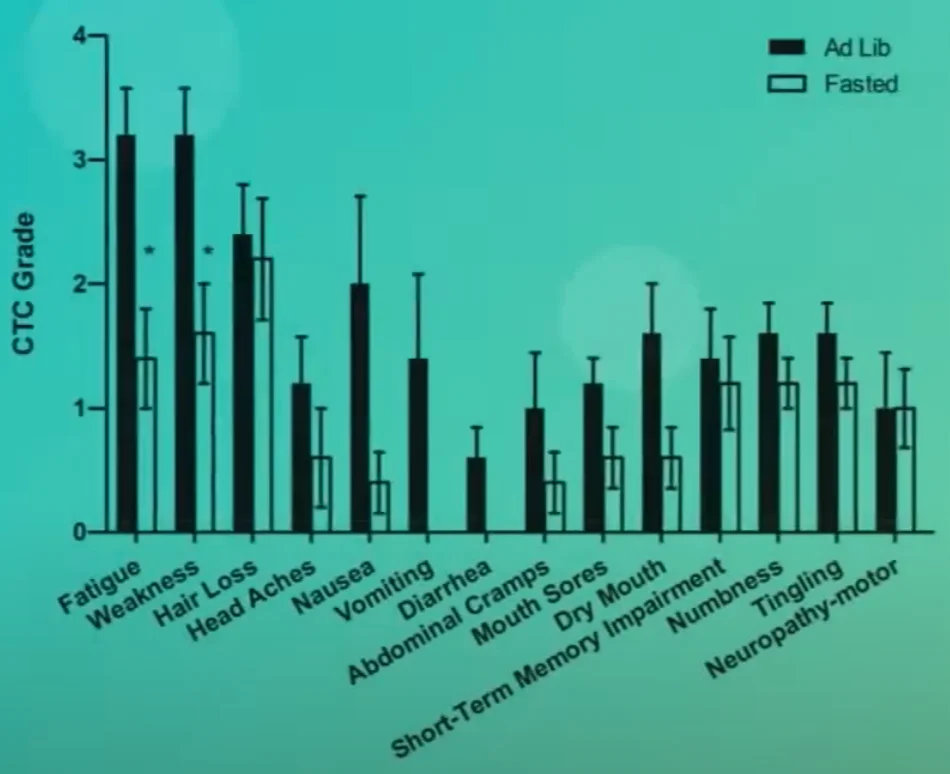
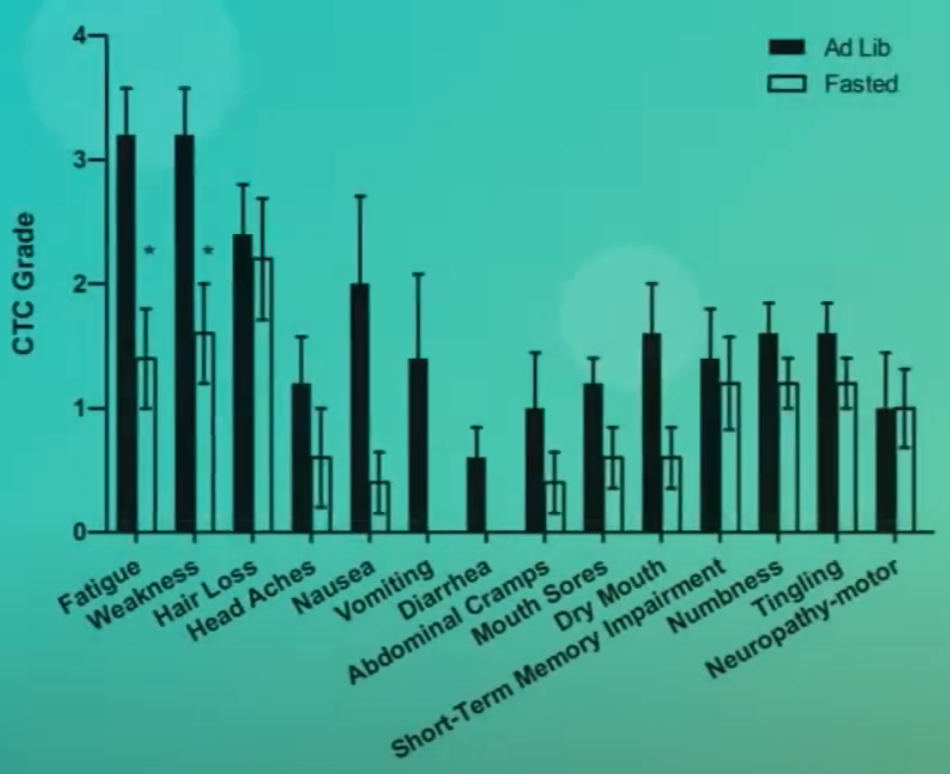
Patient accounts and early clinical studies are beginning to shed light on the human experience with fasting in the context of cancer treatment. Several patients diagnosed with diverse cancers have voluntarily incorporated fasting into their treatment regimens, reporting a notable reduction in common chemotherapy side effects such as fatigue, weakness, and gastrointestinal disturbances. Many described feeling generally better, with some experiencing complete absence of vomiting. The modest weight loss typically incurred during short fasting periods was often quickly regained, and no significant harm was reported, suggesting that fasting, when undertaken under appropriate medical supervision, appears to be safe and potentially beneficial in ameliorating treatment-induced toxicity.
More rigorous scientific inquiry, including randomized clinical trials, is underway. In one study involving breast and ovarian cancer patients, a fasting protocol of 36 hours before chemotherapy and 24 hours afterward appeared to improve patients’ quality of life and reduce fatigue. However, the results have not been uniformly positive. Another study found no significant beneficial effects in terms of overall quality of life, although there was a suggestion of reduced bone marrow toxicity, indicated by higher counts of red blood cells and cells responsible for platelet production. Yet, the expected benefit to white blood cell counts, crucial for immune function, was not observed, leading to disappointment. Researchers speculate that the duration or intensity of the fasting period might have been insufficient in some of these trials.
A comprehensive systematic review analyzing 22 studies offered a broader perspective. It indicated that fasting might not only alleviate chemotherapy side effects, such as organ damage and immune suppression, but could also potentially suppress tumor progression, including growth and metastasis, leading to improved survival outcomes. However, a significant caveat emerged: the vast majority of these studies were conducted on animal models. Human studies were largely confined to evaluating safety and the management of side effects. The critical aspects of fasting’s influence on tumor growth, metastasis, and overall prognosis in humans remain largely unevaluated, highlighting a substantial gap in current knowledge.
Does Fasting Enhance Chemotherapy’s Effectiveness?
The question of whether fasting can make chemotherapy more effective is a central focus of ongoing research. Short-term food withdrawal, particularly for periods like 48 hours before and 24 hours after chemotherapy, is being explored for its potential to mitigate "chemotherapy-induced toxicity." However, the tumor-suppressing effects of fasting itself, beyond its ability to reduce side effects, are still not fully understood or thoroughly evaluated.
Some proponents argue that simply reducing the side effects of chemotherapy could indirectly enhance its efficacy. By allowing patients to better tolerate higher doses of their prescribed medications, the overall therapeutic impact might be amplified. For instance, cardiotoxicity and nephrotoxicity associated with commonly used anti-cancer drugs often limit the achievable therapeutic dose, thereby curtailing their full potential. However, it remains uncertain whether maximizing the tolerated chemotherapy dose would definitively lead to longer survival or an improved quality of life. For the present, the primary focus for many researchers and clinicians remains on the demonstrable benefit of fewer and less severe side effects for their own sake.
Unraveling the Mechanisms: How Does Fasting Work?
Understanding the precise mechanisms by which fasting exerts its effects is crucial for its rational application in cancer care. One significant pathway involves the reduction of Insulin-like Growth Factor-1 (IGF-1). IGF-1 is a potent growth hormone that has been implicated in promoting cancer development and progression. By lowering circulating levels of IGF-1, fasting may create an environment that differentially protects normal cells while rendering cancer cells more vulnerable to chemotherapy. This dual action, protecting healthy tissues while simultaneously hindering tumor growth, presents a promising avenue for therapeutic intervention. Furthermore, modulating IGF-1 signaling might also play a role in cancer prevention.
While several days of fasting can significantly reduce IGF-1 levels by as much as half, this effect is largely attributed to the substantial reduction in protein intake during the fasting period. Protein, particularly animal protein, is identified as a key determinant of circulating IGF-1 levels in humans. This suggests that a reduction in protein intake, especially from animal sources, could become an important component of both anti-cancer and anti-aging dietary strategies.
The Power of Plant-Based Diets in Lowering IGF-1
The role of dietary protein in regulating IGF-1 levels has led to investigations into the impact of different dietary patterns. Comparative studies have highlighted the potent effect of plant-based diets. Individuals adhering to strictly plant-based diets, consuming protein within recommended daily allowances (approximately 0.8 grams per kilogram of body weight), exhibit significantly lower IGF-1 levels compared to individuals of similar build who consume higher amounts of protein, typical of Western diets, even when both groups are calorie-restricted. This suggests that a diet centered around whole plant foods not only down-regulates IGF-1 activity, potentially slowing the aging process, but may also represent a strategy for repurposing anti-aging genetic pathways to combat cancer. The implications are far-reaching, suggesting that dietary choices can profoundly influence the biological environment in ways that may either promote or inhibit cancer development and progression. As research continues to illuminate these complex interactions, fasting and dietary modifications are emerging not as fringe therapies, but as potentially vital components of a comprehensive, evidence-based approach to cancer care.