The intricate architecture of the human gut, a vital organ responsible for nutrient absorption and immune defense, is increasingly understood to be a critical factor in overall health. Recent scientific investigations are shedding light on a phenomenon known as intestinal permeability, or "leaky gut," suggesting it may represent a significant new frontier for both disease prevention and therapeutic intervention. This complex barrier, a marvel of biological engineering, unfurls across an astonishing surface area exceeding 4,000 square feet – a expanse larger than a professional tennis court. Maintaining this vast and crucial interface demands a substantial metabolic investment, consuming approximately 40% of the body’s total energy expenditure.
Growing evidence suggests that the integrity of this intestinal barrier is not merely a passive lining but an active participant in health and disease. Disruptions to this delicate system are being implicated in the pathogenesis of a growing number of conditions, ranging from well-established autoimmune disorders like celiac disease to chronic inflammatory bowel diseases such as Crohn’s disease and ulcerative colitis. The implications of a compromised gut barrier extend beyond digestive ailments, with emerging research linking it to a wider spectrum of chronic diseases.
Understanding Intestinal Permeability: A Scientific Perspective
Intestinal permeability refers to the degree to which the intestinal lining allows substances to pass from the gut lumen into the bloodstream. In a healthy gut, this barrier is highly selective, permitting the passage of digested nutrients while effectively preventing the entry of harmful pathogens, toxins, and undigested food particles. This selective passage is facilitated by specialized junctions between intestinal epithelial cells, known as tight junctions, which act as gatekeepers. When these tight junctions become loose or damaged, intestinal permeability increases, leading to a "leaky gut."
Researchers have employed various methods to assess intestinal permeability. One illustrative technique involves the use of non-absorbable, inert substances like blue food coloring. In individuals with a healthy intestinal barrier, such coloring agents are expected to remain within the gut lumen and be excreted. However, in cases of severe gut barrier damage, these substances can be detected in the bloodstream, indicating a breach in intestinal integrity. A stark example of this was observed in severely ill patients suffering from sepsis, a life-threatening condition characterized by a widespread inflammatory response. In these patients, the blue food coloring was found in their blood, correlating with documented damage to their gut barrier.
It is crucial to understand that developing a leaky gut does not necessitate an ICU admission. The factors contributing to its onset can be far more common and insidious. Even readily available over-the-counter medications, such as aspirin and ibuprofen, have been identified as potential culprits in compromising intestinal barrier function.
The Impact of Common Medications on Gut Health
The widespread use of non-steroidal anti-inflammatory drugs (NSAIDs), including aspirin and ibuprofen, has long been associated with gastrointestinal side effects. Emerging research provides a clearer understanding of the mechanisms behind these adverse effects, directly linking NSAID consumption to increased intestinal permeability.
A notable study demonstrated that ingesting just two standard aspirin tablets (325 mg each) or two extra-strength aspirin tablets (500 mg each) in a single dose was sufficient to significantly increase gut leakiness. These findings suggest a cautionary approach for all individuals, even those who consider themselves healthy, regarding the use of aspirin. The drug’s capacity to induce gastrointestinal barrier dysfunction warrants careful consideration of its necessity and potential risks.
Concerns have also been raised about buffered aspirin, a formulation that combines aspirin with an antacid, ostensibly to mitigate gastrointestinal irritation. However, scientific evidence suggests this buffering effect may be illusory. Studies employing endoscopic examinations have revealed that both regular aspirin and buffered aspirin formulations can lead to the formation of multiple erosions in the delicate inner lining of the stomach and intestines. In a study where researchers visually inspected the gastrointestinal tracts of individuals who had taken aspirin or buffered aspirin at recommended doses, approximately 90% exhibited significant erosions and redness. This damage can occur with remarkable speed, with evidence suggesting it can manifest within a mere five minutes of ingestion.
In contrast, acetaminophen, widely known by its brand name Tylenol in the United States, appears to pose a lower risk for direct gastrointestinal damage. While it is generally considered a safer alternative for pain relief in the absence of liver issues, it is essential to acknowledge its potential limitations and contraindications. Interestingly, the role of vitamin C, often promoted for its health benefits, has also come under scrutiny in this context. Rather than offering protection, some research indicates that vitamin C supplements may actually exacerbate aspirin-induced increases in gut leakiness.
The implications of NSAID-induced gut leakiness extend to allergic reactions. There is a recognized association between NSAID use and food-induced anaphylaxis. These drugs are implicated in up to 25% of such severe allergic reactions, increasing the odds of life-threatening food allergy attacks by more than tenfold. The prevailing hypothesis is that NSAIDs compromise the intestinal barrier, allowing minuscule food particles to permeate the gut lining and enter the bloodstream. This premature entry can trigger an exaggerated immune response, leading to anaphylaxis in susceptible individuals.
Beyond Medications: Lifestyle Factors Contributing to Leaky Gut

While medications are significant contributors, other common lifestyle elements can also impact intestinal barrier function.
The Strenuous Exercise Paradox
Even seemingly beneficial activities like strenuous exercise can, under certain circumstances, transiently affect gut integrity. Prolonged periods of intense physical exertion, such as an hour at 70% of maximum capacity, can divert a substantial amount of blood flow away from internal organs, including the intestines, towards the working muscles. This physiological shift can lead to temporary ischemia and mild gut leakiness. The situation can be further exacerbated if athletes concurrently use NSAID drugs, a practice that is unfortunately prevalent. This combination of strenuous exercise and NSAID use presents a heightened risk for compromised gut health.
Alcohol Consumption and Gut Permeability
Alcohol, a widely consumed beverage, has also been identified as a risk factor for food allergy attacks, primarily due to its capacity to increase gut leakiness. The mechanism is similar to that of NSAIDs: alcohol can weaken the intestinal barrier, allowing undigested food particles to enter the bloodstream and trigger adverse immune responses. Fortunately, the gut’s capacity for healing is significant, and abstaining from alcohol can often lead to a restoration of intestinal barrier function.
Dietary Fat and the Gut Microbiome
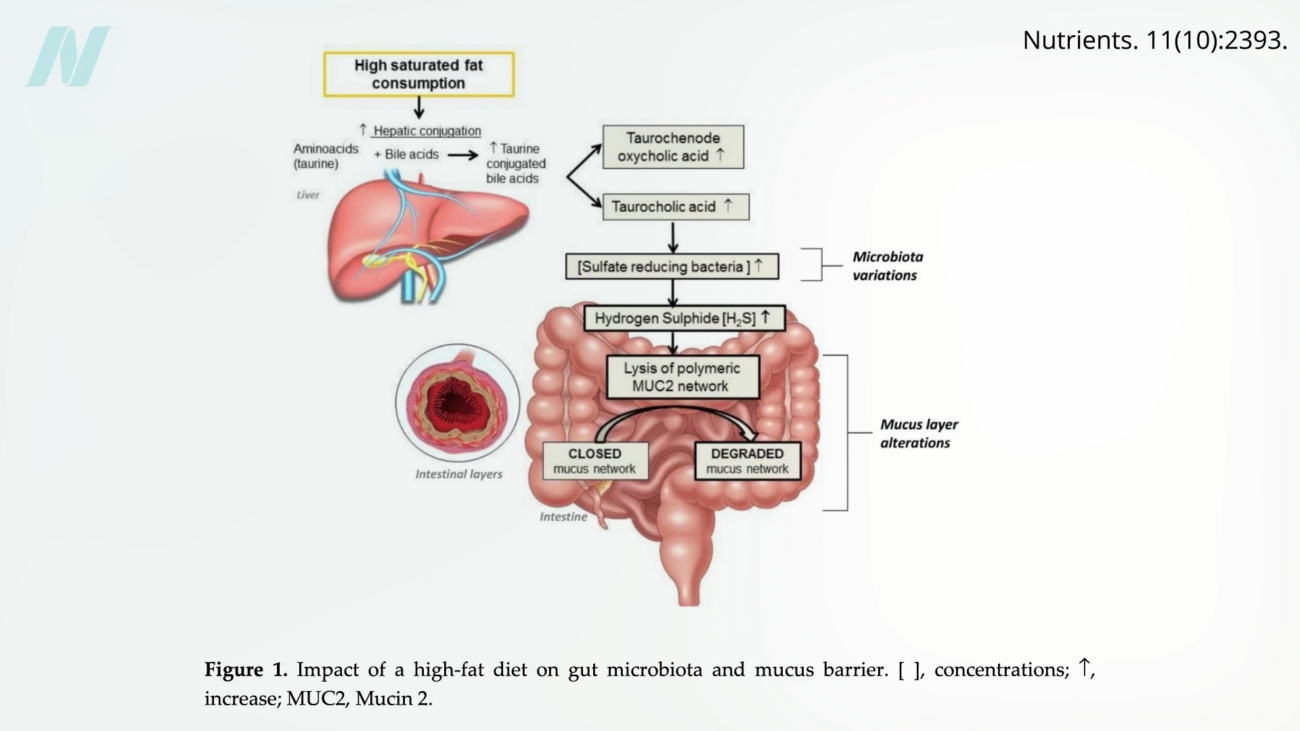
Dietary patterns play a pivotal role in shaping the health of the intestinal barrier and the resident gut microbiome. Elevated consumption of saturated fats, commonly found in red meat, full-fat dairy products, and processed "junk" foods, has been linked to detrimental changes in gut health. Saturated fats can promote the growth of certain bacteria that produce hydrogen sulfide, a gas with a rotten-egg odor. Hydrogen sulfide, in excessive amounts, can degrade the protective mucus layer that lines the intestinal wall, leaving the underlying epithelium vulnerable.
The broader impact of high-fat diets on intestinal health is a subject of extensive research. While many studies have historically been conducted on laboratory animals or in vitro, the results consistently point to a negative impact on the intestinal barrier. The concern is whether these findings translate to human physiology.
The parallel rise in obesity and cardiometabolic disorders alongside a global dietary shift from traditional lower-fat to higher-fat patterns has prompted closer examination of this correlation. Disturbances in the balance of beneficial gut bacteria, known as dysbiosis, have been strongly associated with an increased risk of many of these chronic diseases. Rodent studies have indicated that high-fat diets can disrupt the gut microbiome and impair the gut barrier, ultimately contributing to disease development.
To bridge the gap between animal studies and human health, researchers have conducted human interventional trials. A notable six-month randomized controlled feeding trial investigated the effects of dietary fat on the gut microbiota. The findings corroborated earlier suspicions: higher fat consumption was indeed associated with unfavorable alterations in the gut microbiome and an increase in pro-inflammatory markers in the blood. Importantly, this study did not exclusively focus on saturated fats from animal products. Instead, it demonstrated that simply replacing refined carbohydrates with refined fats – for instance, swapping white rice and wheat flour for soybean oil – could elicit these negative changes. These results carry significant implications for public health messaging, suggesting that countries undergoing dietary transitions towards Westernized diets should advise against increasing fat intake, while those that have already adopted such diets should consider strategies for reduction.
Broader Implications and Future Directions
The growing body of evidence linking intestinal permeability to a wide array of health conditions underscores the critical importance of maintaining a healthy gut barrier. This understanding opens new avenues for therapeutic interventions, potentially targeting the gut lining itself to prevent or manage chronic diseases.
The research into leaky gut is not without its complexities. While significant progress has been made, further human-based, long-term studies are essential to solidify these findings and develop precise therapeutic strategies. The interplay between diet, lifestyle, medications, and the gut microbiome is intricate, and a holistic approach is likely to be most effective.
As scientific understanding evolves, the focus is shifting towards proactive measures that support intestinal barrier integrity. Dietary modifications, judicious use of medications, and mindful lifestyle choices are emerging as powerful tools in the prevention and management of conditions associated with leaky gut. The ongoing exploration of this fascinating area of human physiology promises to unlock new insights into health and disease, paving the way for innovative approaches to well-being.
Doctor’s Notes on Leaky Gut Management
For individuals seeking to understand and address leaky gut concerns, further exploration of related topics is recommended. The theory connecting animal product consumption to inflammation via a "leaky gut" mechanism warrants attention, as does the potential for dietary interventions to promote gut healing. Discussions surrounding Small Intestinal Bacterial Overgrowth (SIBO) also often intersect with the concept of gut leakiness, highlighting the interconnectedness of these gastrointestinal health issues. Comprehensive resources are available to guide individuals in making informed decisions about their gut health and overall well-being.