The delicate lining of our intestines, a critical yet often overlooked component of human health, is under constant siege from everyday substances, according to emerging scientific research. This protective barrier, responsible for absorbing nutrients while preventing harmful substances from entering the bloodstream, can become compromised, a condition colloquially termed "leaky gut" or medically known as increased intestinal permeability. This disruption is no longer considered a fringe theory but a potential nexus for disease prevention and therapeutic intervention, impacting conditions ranging from autoimmune disorders to life-threatening allergic reactions.
The Vast and Vital Intestinal Barrier
The sheer scale of the intestinal barrier underscores its significance. Composed of a single layer of epithelial cells tightly bound together, it forms an intricate landscape of folds and villi, dramatically expanding the surface area available for nutrient absorption. This vast expanse, estimated to be over 4,000 square feet – larger than a tennis court – is a powerhouse of biological activity. Maintaining this complex structure demands a substantial portion of the body’s energy resources, accounting for approximately 40% of total energy expenditure. This high metabolic demand highlights the immense importance placed by the body on the integrity of this vital barrier.
Unraveling the Mechanisms of Intestinal Permeability
Recent scientific investigations are increasingly pointing to the disruption of this intestinal barrier as a key factor in the development of a spectrum of chronic and acute conditions. Conditions such as celiac disease, an autoimmune disorder triggered by gluten, and inflammatory bowel disease (IBD), which encompasses conditions like Crohn’s disease and ulcerative colitis, have been strongly linked to compromised gut barrier function.
Researchers have employed various methodologies to assess intestinal permeability. One notable approach involves the use of inert marker substances, such as blue food coloring, which can be ingested and then tracked. In healthy individuals, these markers are largely contained within the intestinal lumen, reflecting a robust and intact barrier. However, in individuals with severely compromised gut integrity, such as those suffering from sepsis – a life-threatening systemic inflammatory response to infection – these markers have been detected in the bloodstream. This observation starkly illustrates the functional consequences of a damaged intestinal barrier, where normally contained substances can translocate into circulation.
Common Culprits: Pharmaceuticals and Their Impact
The notion that everyday medications can contribute to a "leaky gut" is a significant public health concern. Non-steroidal anti-inflammatory drugs (NSAIDs), a ubiquitous class of pain relievers and anti-inflammatory agents, have been identified as particularly problematic. The evidence suggests that even a single dose of common NSAIDs can significantly increase intestinal permeability.
For instance, studies have demonstrated that taking just two standard 325 mg aspirin tablets, or two extra-strength 500 mg tablets, can measurably increase gut leakiness. This finding is particularly concerning for the general population, as it implies that even individuals without underlying health conditions could be inadvertently compromising their intestinal barrier function through routine pain management. The implications extend to the widespread use of these medications for conditions like headaches, menstrual cramps, and minor aches and pains.
Even buffered aspirin, marketed as a gentler alternative due to its antacid component intended to mitigate gastrointestinal irritation, has been found to offer little protection against the damaging effects on the intestinal lining. Research involving endoscopic examinations of individuals who took aspirin or buffered aspirin at recommended doses revealed extensive erosions and redness within the stomach and intestinal lining in a significant percentage of participants. Disturbingly, these erosions can manifest rapidly, with evidence suggesting damage can occur within five minutes of ingestion.
In contrast, acetaminophen, commonly known as Tylenol, appears to present a lower risk for direct gastrointestinal damage, making it a potentially safer alternative for pain relief, provided individuals do not have pre-existing liver conditions. Interestingly, the role of vitamin C supplements in this context has also been examined. Contrary to potential expectations, some research suggests that vitamin C supplementation may exacerbate aspirin-induced increases in gut leakiness, further complicating the landscape of dietary and supplemental interventions.
NSAIDs and the Escalation of Food Allergies
The disruption of the intestinal barrier by NSAIDs like aspirin, ibuprofen, and naproxen may also play a role in the increased prevalence and severity of food allergies. Scientific literature indicates that NSAID use is associated with a significant portion of food-induced anaphylaxis cases. Specifically, individuals taking these medications exhibit a more than tenfold increased risk of life-threatening allergic reactions to food. The proposed mechanism involves the increased intestinal permeability caused by NSAIDs, allowing undigested food particles to "leak" into the bloodstream, where they can trigger a robust immune response and potentially lead to anaphylaxis. This connection highlights a critical interplay between medication use and immune system dysregulation.
The Physical Demands of Exercise and Gut Health
While exercise is widely lauded for its health benefits, the intensity and duration of physical activity can also influence intestinal barrier function. Strenuous exercise, particularly prolonged bouts at high intensity (e.g., an hour at 70% of maximum capacity), can divert blood flow away from internal organs, including the intestines, to support working muscles. This physiological shift can lead to transient injury to the intestinal lining, resulting in mild gut leakiness.
The problem is compounded when athletes combine strenuous exercise with NSAID use. The synergistic effect of reduced blood flow and the direct damaging impact of NSAIDs can significantly aggravate gut leakiness, potentially increasing the risk of gastrointestinal distress and other complications. This underscores the importance of careful consideration of medication use among athletes, especially during periods of intense training.

Alcohol’s Contribution to Gut Permeability and Allergic Responses
Alcohol consumption has also been identified as a risk factor for food allergy attacks, operating through a similar mechanism of increasing gut leakiness. By compromising the integrity of the intestinal barrier, alcohol can facilitate the passage of food particles into the bloodstream, potentially triggering or exacerbating allergic reactions. The good news, however, is that abstaining from alcohol can allow the gut to heal, suggesting a direct pathway to recovery through dietary and lifestyle modifications.
Dietary Fat and the Gut Microbiome: A Complex Relationship
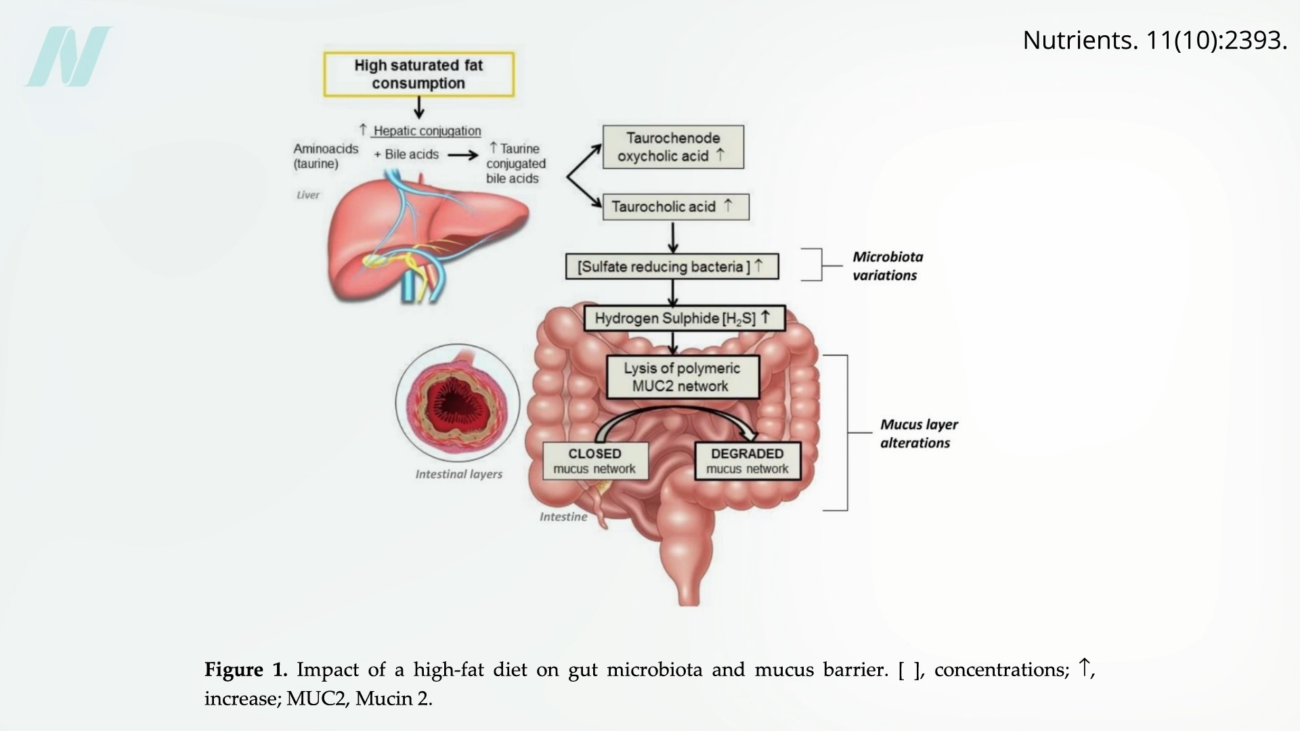
Beyond pharmaceuticals and lifestyle choices, dietary components play a pivotal role in maintaining intestinal health. Elevated consumption of saturated fats, commonly found in red meat, dairy products, and many processed or "junk" foods, has been linked to detrimental changes in the gut environment. Saturated fats can promote the growth of certain bacteria that produce hydrogen sulfide, a gas with a characteristic rotten-egg smell. Hydrogen sulfide, at elevated levels, can degrade the protective mucus layer that lines the intestines, further compromising the barrier.
The broader impact of high-fat diets on intestinal health is a subject of extensive research. While many studies have focused on animal models or in vitro experiments, the human relevance is becoming increasingly clear. A growing body of evidence suggests that high-fat diets negatively affect the intestinal barrier through various mechanisms.
The Rise of Western Diets and Cardiometabolic Disorders
The global increase in obesity and other cardiometabolic disorders, such as type 2 diabetes and heart disease, has coincided with a widespread dietary transition from traditional, lower-fat eating patterns to higher-fat, Westernized diets. This shift has been accompanied by significant alterations in the gut microbiome, the complex community of microorganisms residing in the intestines. Disturbances in the balance of beneficial and harmful gut bacteria have been associated with an elevated risk of many of these same chronic diseases.
Rodent studies have provided compelling insights into the effects of high-fat diets on the microbiome and gut barrier. These studies indicate that such diets can unbalance the microbial ecosystem and impair gut barrier function, ultimately contributing to disease development. To bridge the gap between animal studies and human health, robust human interventional trials are essential.
Human Trials Confirm the Impact of Dietary Fat
A notable six-month randomized controlled feeding trial provided crucial human data on the effects of dietary fat on the gut microbiota and barrier function. This trial revealed that higher fat consumption was indeed associated with unfavorable changes in the gut microbiome and an increase in proinflammatory factors circulating in the blood. Importantly, this study did not solely focus on saturated fats; rather, it demonstrated that simply replacing refined carbohydrates with refined fats – for example, substituting soybean oil for white rice and wheat flour – could lead to these adverse effects.
These findings have significant public health implications. They suggest that countries undergoing dietary transitions towards Westernized patterns should actively discourage an increase in dietary fat intake. For populations that have already adopted such diets, a reduction in overall dietary fat consumption may be a critical strategy for improving gut health and mitigating the risk of associated chronic diseases.
Implications for Public Health and Future Research
The accumulating evidence paints a clear picture: the integrity of the intestinal barrier is a critical determinant of overall health, and its disruption can have far-reaching consequences. From common medications to dietary choices and even intense physical activity, numerous everyday factors can compromise this vital defense system.
The implications for disease prevention are substantial. By understanding and addressing the factors that contribute to a leaky gut, interventions can be developed to strengthen the intestinal barrier and potentially prevent the onset or progression of various chronic diseases. This includes encouraging mindful medication use, particularly NSAIDs, and promoting dietary patterns that support gut health, such as those rich in fiber and lower in saturated and refined fats.
Future research will likely continue to explore the intricate mechanisms by which these various factors influence intestinal permeability and the gut microbiome. Further human interventional trials are needed to refine our understanding of optimal dietary strategies and to develop targeted therapies for conditions associated with increased intestinal permeability. The concept of the "leaky gut" is evolving from a theoretical concern to a tangible target for improving human health and well-being.
Doctor’s Notes: Addressing Gut Health
For those seeking to delve deeper into the complexities of gut health and the role of diet, further resources are available. Discussions surrounding the "leaky gut theory" and its potential link to inflammation caused by animal product consumption offer valuable insights. Additionally, actionable guidance on how to heal a leaky gut through dietary adjustments provides practical strategies for individuals looking to improve their intestinal health. Information on managing conditions like Small Intestinal Bacterial Overgrowth (SIBO), which can be influenced by gut permeability, also highlights the interconnectedness of these gastrointestinal health issues.