Recent investigations into the efficacy of medical procedures, particularly through the use of "sham surgery" trials, have brought into question the effectiveness and safety of some widely adopted surgical interventions. These studies, designed to rigorously test the true benefits of surgical techniques by comparing them against a placebo or simulated procedure, have yielded startling results, suggesting that in some cases, the perceived advantages of popular surgeries may be minimal or even non-existent when stripped of psychological factors and the placebo effect.
The Intragastric Balloon: A History of Hype and Hazard
The intragastric balloon, introduced in the 1980s, was heralded as a groundbreaking, minimally invasive solution for weight loss. The concept was straightforward: a deflated balloon was inserted into the stomach and then inflated with air or water, occupying a significant portion of the gastric space. This was intended to create a feeling of fullness, thereby reducing food intake and promoting weight loss. However, the trajectory of the intragastric balloon’s journey into widespread use mirrors a recurring concern within the medical device industry – the premature market entry of products lacking comprehensive evidence of their long-term efficacy and safety.
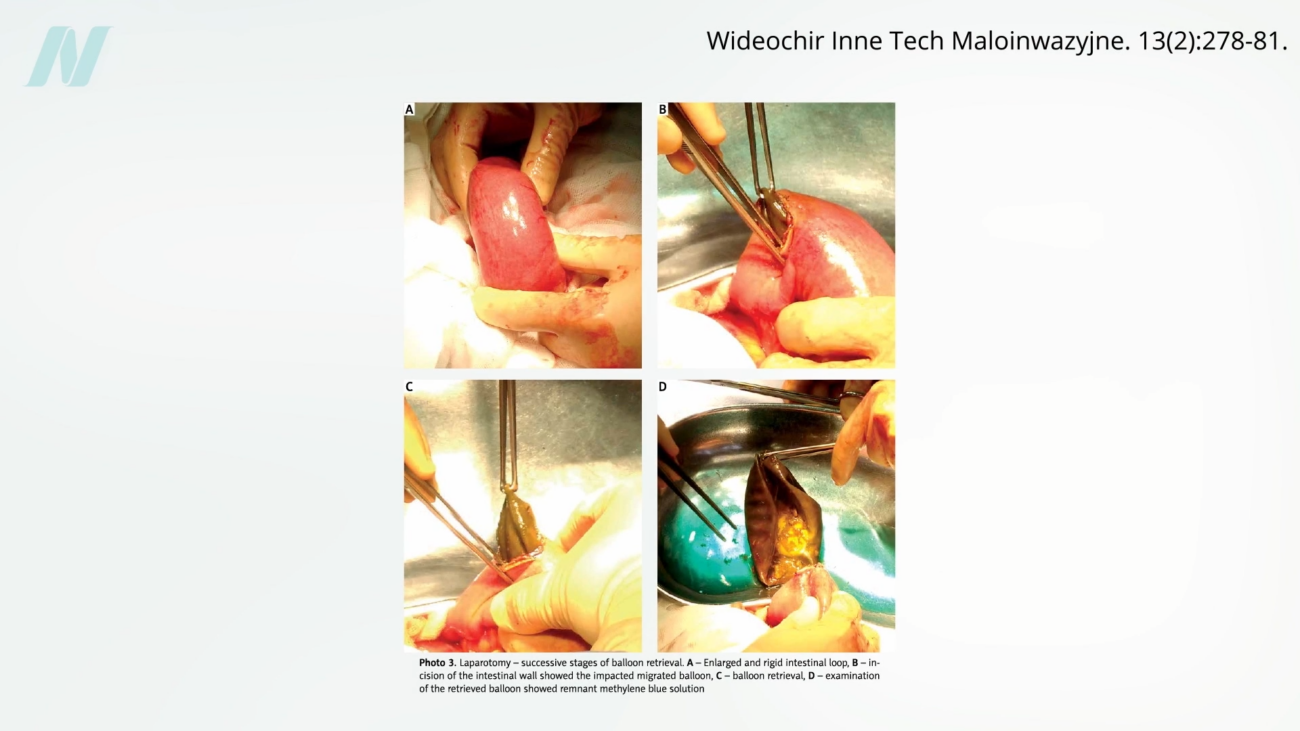
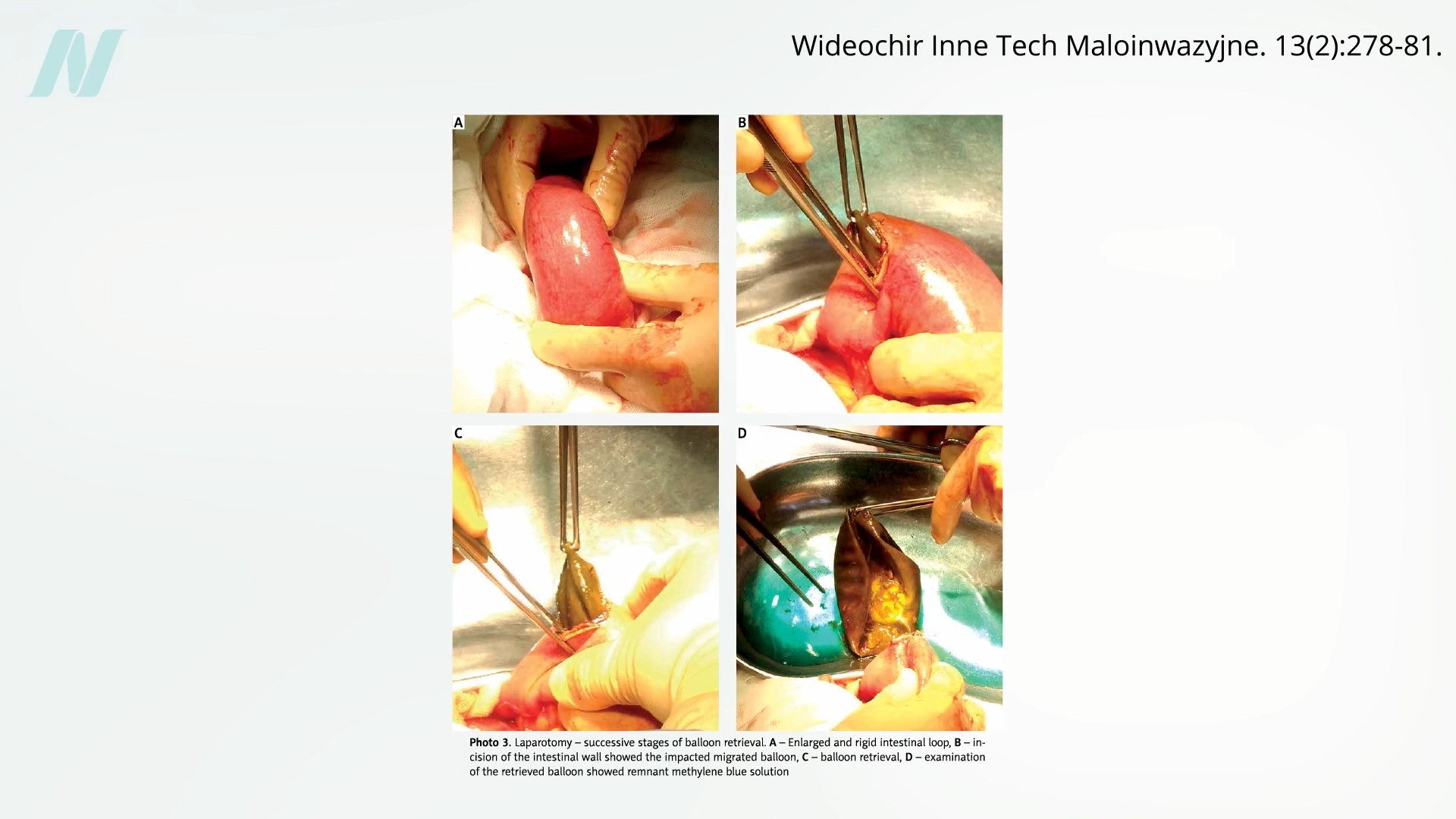
Early clinical experiences with these balloons quickly revealed significant shortcomings. A pivotal study conducted at the Mayo Clinic brought the "gastric bubble" concept under intense scrutiny. The findings were concerning: a staggering eight out of every ten intragastric balloons spontaneously deflated. This mechanical failure posed a direct and serious risk to patients. A deflated balloon could migrate into the intestines, leading to a potentially life-threatening bowel obstruction. Visual evidence from medical imaging and patient case studies has starkly illustrated the dangers of such complications.
Beyond the immediate risk of mechanical failure, the balloons also demonstrated a concerning propensity for causing gastrointestinal damage even before deflation. Reports indicated that up to half of patients experienced gastric erosions, signifying damage to the stomach lining. The most damning finding, however, was that these devices failed to provide a significant weight loss advantage when directly compared to non-surgical interventions like dietary changes and behavioral modification strategies. Despite these early red flags, intragastric balloons were eventually withdrawn from the market.

The Return and Re-evaluation of Intragastric Balloons
After a hiatus of over three decades, intragastric balloons made a resurgence in the United States. In 2015, the U.S. Food and Drug Administration (FDA) began approving a new generation of these devices. Within a short period, more than 5,000 of these newer balloons were implanted. This reintroduction coincided with the passage of the Sunshine Act, a landmark piece of legislation designed to increase transparency in the financial relationships between the healthcare industry and medical professionals. The Act mandates the disclosure of payments made by pharmaceutical companies and medical device manufacturers to physicians, shedding light on potential conflicts of interest.
While the public has become increasingly aware of the financial ties between doctors and "Big Pharma," the extent to which surgeons may receive payments from companies for the medical devices they use is a less widely understood issue. Data revealed by the Sunshine Act has shown that some top physicians receive substantial financial compensation from device companies. In one instance, the top 100 physician recipients of industry payments garnered an astounding $12 million from device companies in a single year. Alarmingly, a significant portion of these physicians failed to disclose these financial conflicts of interest when publishing research related to the devices they used, raising serious questions about the objectivity of their findings.
While modern intragastric balloons are designed to be reversible, a characteristic that distinguishes them from more permanent bariatric surgical procedures, their safety profile remains a significant concern. The FDA has issued multiple advisories highlighting the potential risks associated with these devices, including the possibility of death. The mechanisms by which such severe complications can occur are often counterintuitive. For example, a smooth, rounded object like a balloon can, paradoxically, lead to a fatal stomach perforation. This can happen when the balloon induces severe, persistent nausea and vomiting, leading to such forceful retching that the stomach wall ruptures. Nausea and vomiting are reported as very common side effects, affecting the majority of patients. Furthermore, persistent vomiting can contribute to life-threatening nutrient deficiencies following balloon implantation.
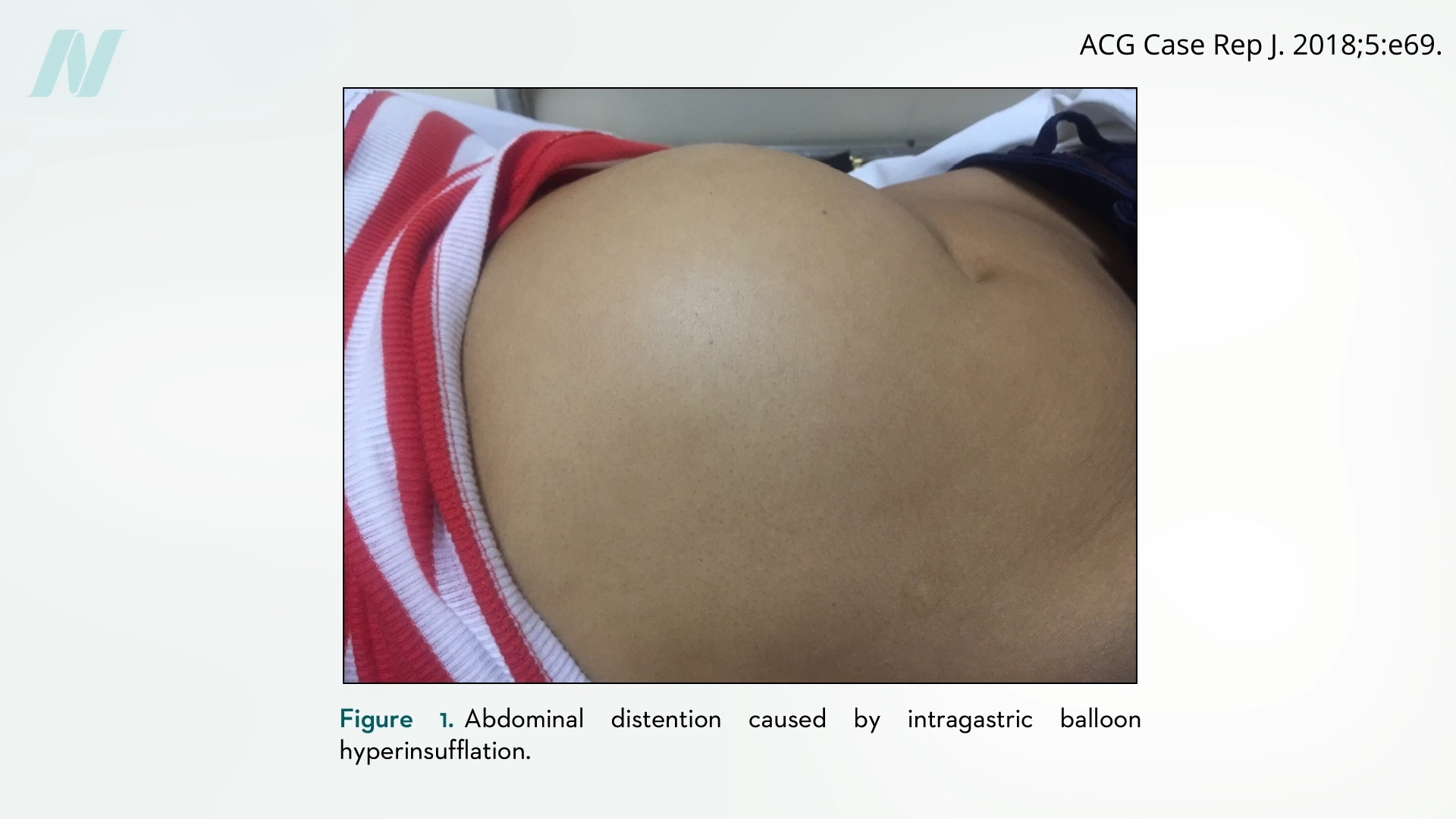
Complications with these devices are not limited to deflation. Some adverse events, such as bowel obstructions, are indeed linked to the balloons deflating and migrating. However, a peculiar and concerning issue has also emerged: the spontaneous overinflation of balloons. This phenomenon can lead to severe pain, vomiting, and abdominal distention, mirroring the symptoms of a deflated balloon causing an obstruction. This issue has drawn parallels to complications observed with breast implants, where a phenomenon known as "spontaneous autoinflation" has been documented, leading to an unintended and significant increase in breast volume. Early experimental intragastric balloons even utilized technology originally developed for breast implants, underscoring a historical link between these device types and unpredictable behavior.
The Rise of Sham Surgery as a Scientific Tool
The concept of "sham surgery" has emerged as a critical methodology for disentangling the true physiological effects of a surgical procedure from the powerful influence of the placebo effect and patient expectation. In clinical trials, particularly those involving interventions where a direct comparison to a non-intervention is ethically or practically challenging, sham surgery provides a robust control group. In such trials, participants are randomly assigned to receive either the actual surgical procedure or a simulated surgery. The sham procedure is designed to mimic the actual surgery as closely as possible, including incisions and even the sounds and sensations associated with the procedure, but without performing the therapeutic intervention itself. For example, in a knee surgery trial, a sham surgery might involve making an incision and simulating the act of debridement or arthroscopy without actually operating on the joint.
Testing the Efficacy of Knee Surgery: A Landmark Study
A seminal study published in 2002 in The New England Journal of Medicine dramatically illustrated the power of sham surgery in evaluating the efficacy of common medical procedures. The focus was on arthroscopic knee surgery, one of the most frequently performed orthopedic procedures globally, with billions of dollars spent annually on interventions for osteoarthritis and knee injuries. The study randomized patients suffering from knee pain into two groups: one receiving the actual arthroscopic surgery, and the other undergoing a sham procedure. The sham group experienced incisions and simulated surgical actions, including the introduction of saline to mimic irrigation, but their joints were left untreated.
The results of this trial sent shockwaves through the medical community and sparked considerable ethical debate. Professional medical associations questioned the ethics of subjecting patients to sham surgery, and the sanity of patients who consented to participate was also brought into question. However, the findings were undeniable: patients in both the actual surgery group and the sham surgery group experienced significant improvements in their knee pain. Crucially, the study demonstrated that the observed improvements in the surgical group were not statistically superior to those in the placebo group, suggesting that the perceived benefits of arthroscopic knee surgery were largely attributable to the placebo effect, patient expectations, and the natural course of healing. This groundbreaking study cast doubt on the necessity and effectiveness of this widely performed surgery.
Broader Implications and Industry Influence
The implications of sham surgery trials extend far beyond the specific procedures examined. They highlight a broader challenge in medical research: the difficulty in isolating the true therapeutic benefit of an intervention from psychological factors and the inherent trust patients place in medical professionals and procedures. The financial incentives within the medical device industry, as illuminated by the Sunshine Act, further complicate this picture. When surgeons receive significant payments from device manufacturers, there is a potential for bias in the research they conduct and the procedures they advocate for.
Industry-funded trials often report "notable weight loss" for interventions like intragastric balloons. However, critics argue that it is difficult to discern the actual effect of the device itself from the accompanying supervised diet and lifestyle changes that are integral to these study protocols. The challenge of eliminating the placebo effect in surgical trials is substantial, as patients undergoing a procedure, even a sham one, have higher expectations of improvement compared to those receiving non-surgical treatments.
Current research continues to explore the efficacy of other popular surgical interventions. Rotator cuff shoulder surgery, for instance, is currently facing a similar crisis of confidence, with ongoing investigations seeking to determine its true benefit beyond the placebo effect.
When intragastric balloons were subjected to sham-controlled trials, the results were consistent with earlier findings: both older and newer devices often failed to demonstrate any significant weight-loss benefit. Even in cases where some weight loss was observed, it was often temporary. Balloons are typically allowed to remain in place for only six months due to the increasing risk of deflation. Attempts to extend this duration or to use sequential balloon placements have not improved long-term weight outcomes. Sham-controlled trials have indicated that any effects of the balloon on appetite and satiety can diminish over time as the body adapts.
The overarching message from sham surgery trials is a sobering one: some of the most frequently performed and widely accepted surgical procedures may offer little to no genuine therapeutic benefit beyond the placebo effect. This challenges the traditional self-perception of physicians as purely scientific practitioners and raises concerns about the potential for medical practices to be influenced by factors other than rigorous evidence. The medical community’s strong stance against movements that reject established scientific consensus, such as anti-vaccination sentiments, stands in stark contrast to the findings that some of its own popular interventions may be based on flawed premises or "alternative facts." The progression of osteoarthritis patients who undergo knee surgery to requiring total knee replacement, as noted in some research, further underscores the potential for interventions to exacerbate rather than alleviate underlying conditions.
Looking Ahead: Sustainable Weight Loss and Ethical Considerations
The insights gained from sham surgery trials underscore the importance of evidence-based medicine and critical evaluation of all medical interventions. For individuals seeking sustainable weight loss, the focus must remain on proven, non-surgical strategies that address diet and lifestyle. This ongoing discussion also highlights the ethical imperative for complete transparency in medical research and practice, particularly concerning financial relationships between healthcare providers and industry. As the medical field continues to evolve, a commitment to rigorous scientific inquiry and patient-centered care, unclouded by commercial interests, is paramount.
Doctor’s Note:
The subsequent installment in this series will delve into "Extreme Weight-Loss Devices," exploring their efficacy and safety profiles. Further information on bariatric surgery and related topics can be found in related posts and in the author’s book, How Not to Diet, which is dedicated to sustainable weight loss strategies. Proceeds from book sales are donated to charity.